Multi-tasking protein: destabilizing the cellular system?
Not all proteins need to fold into a single, well defined 3D structure to become functional. Some proteins seem to defy the structure function paradigm. These so called intrinsically disordered proteins (IDPs) are believed to adopt specific conformations and functions upon interaction with different partners in an interaction network. The IDP alpha-synuclein is abundantly expressed in neurons. Its physiological function is not well understood but the aggregation of alpha-synuclein into amyloid fibrils is associated with Parkinson’s disease. Here you will investigate the alpha-synuclein interaction network in the cell.
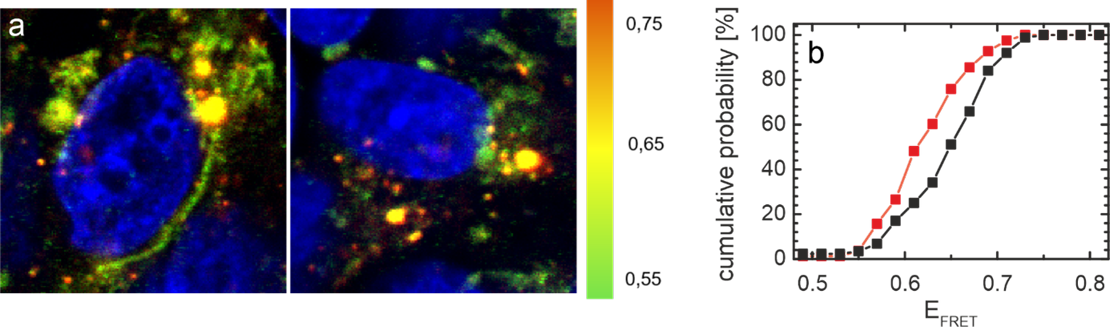
Approach: You will use quantitative colocalization microscopy to gain insights into the alpha-synuclein interaction network in the cell. In a second step you will explore what happens if you disturb the interaction network by e.g. inhibiting interactions or by introducing new interaction partners.
The work can include:
- · Cell culture.
- · Immunostaining.
- · Introduction of transient/inducible expression of additional proteins into cells.
- · Quantitative confocal fluorescence microscopy.
- · Characterization of the interactions in vitro e.g. by FCS or MST.
If you are interested, please feel free to contact us. Within the Nanobiophysics group we try to provide you with a BSc/MSc assignment that fits your expertise and interest.
Please contact:
Mireille Claessens: m.m.a.e.claessens@utwente.nl, Zuidhorst 163
Christian Blum: c.blum@utwente.nl, Zuidhorst 168
