Introduction
My group and I interface supramolecular chemistry and materials science with cell biology and regenerative medicine. We work on dynamic chemical strategies to control physico-chemical properties of biointerfaces, which is key to modulate cell behavior on material surfaces for the design of high-performance biomaterials, cell-tissue constructs and advanced culturing systems.
While the ability to map biochemical signaling within cell junctions has advanced, physico-chemical cues remain difficult to control in space, time and dynamcis. My group has developed biointerfaces with dynamic complexity in combination with lithographic and microscopic technologies.
We use these past achievements to further engage into cellular applications to mimic and perturb cell-cell and cell-extracellular matrix junctions, to study mechanotransduction, cell adhesion/spreading/differentiation with unprecedented precision in space, time and dynamics. We derive and predict global variables of cell responses to ligand dynamics, and introduce them into 3D-environments to widen future biomedical applicability.
Recent key group contributions:
(i) New (non)covalent oriented peptide/protein anchoring strategies and validated them for retention of structure and function of the anchored proteins.
(ii) First introduction of host-guest motifs into the crowded complexity of bacterial and cellular membranes.
(iii) First-time demonstrations of host-guest based photo-/electroactive modulation of peptide and protein assembly and cell adhesion on interfaces.
(iv) New gradient formation procedures, used for threshold binding studies.
(v) Increased kinetic/thermodynamic understanding of supramolecular interactions at surfaces.
(vi) Achieved air-stable, non-fouling and cell-instructive lipid bilayer coatings on biomaterials with control over ligand density and ligand mobility.
(vii) In-vivo validated cell-instructive growth factor interfaces.
Our laboratories are equipped with synthetic and analytical labs, cell culture and bacteriologic labs and cleanrooms. We have access to characterization, purification and synthetic equipment: Jeol AccuTOF (JMS-T100LC), Veeco AFM (nanoscope III/IV), Jasco CD spectrophotometer, Peptide synthesizer (µwave), NTegra Confocal/AFM microscope, Contact Angle, Elemental analysis, Electrochemisty, PE Fluorimeter, PE UV/vis, FT-IR with PM-IRRAS, GC, HPLC, FPLC, Maldi-ToF, Nanoimprint, NMR 400, 600 MHz, Oxygen, Argon cleaner, DLS, SPR.
We are active in the bachelor and master programs of Chemical Engineering and Biomedical Technology and we host with pleasure, at any time convenient, bachelor and master students for final projects. Please write to p.jonkheijm@utwente.nl for project descriptions.
Research topics
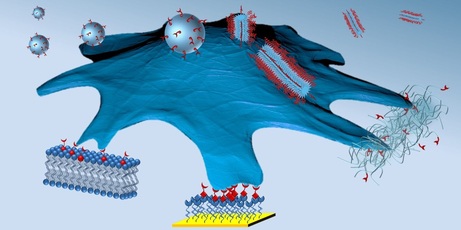
Dynamic supramolecular coatings for biomedical applications
Supramolecular host-guest systems deal with molecules that interact through non-covalent forces. By careful design and selection of the host and guest molecules, we are able to make bio-mimetic coatings that assemble in an organized manner on surfaces for implants, biosensors and organ-on-a-chip devices. These coatings can also be designed to respond to external stimuli, like light and electricity, thus altering their characteristics when required. Currently our focus lies on making coatings that target proteins, growth factors, bacteria and eukaryotic cells.
Bio-functional artificial membranes
Artificial cell membranes in the form of planar supported lipid bilayers (SLBs) are self-assembled structures that mimic the cell membrane while retaining their physicochemical properties (i.e. phase behavior). SLBs can be bio-activated through various supramolecular interactions using biotin conjugates, lipidated peptides or ternary host-guest complexes to engineer biomimetic platforms. These platforms allow us to dynamically interface cells with biomolecules designed for specific interactions and subsequently study biological processes. These artificial cell membranes can be applied on different supports like glass, bio-materials and polymers. Further spatial distribution of ligands on a nano-scale level is made possible by creating gradients in our chip-based device as well as by exploiting domain formation.
Multivalent bioactive ligands
Multivalency is a fundamental principle governing various biological processes in our body. A challenge faced by nanomedicine today is to design multivalent ligands, which can mimic ligand-receptor binding on the cellular level. In our group we are trying to explore various strategies to develop multivalent ligands and use them for studying key bio-signaling pathways responsible for organogenesis.
Chip-printing technologies for proteins
Cell membranes are covered with multiple receptors and other biomolecules that play a pivotal role in transferring signals between cells and the external environment. In order to achieve a deeper understanding of these dynamic cellular processes we aim to replicated cell membranes on a multiplexed arrays using microcontact printing and nanoimprint technology. We develop chips that contain miniscule circuits and patterns of surface-bound peptides and proteins that enables to determine exactly how the biomolecules on the chips communicate with living cells. Immobilization of the biomolecules follows bio-orthogonality and specifity.
