Bioartificial pancreas devices for treatment of type I diabetes
In 2017, Diabetes type 1 (T1D) and type 2 (T2D) are responsible for more that 5 million deaths world-wide. Annually one in ten of adult deaths in Europe is related to diabetes. T1D is characterized by a high glucose level in the blood and an absolute insulin deficiency. Unfortunately, current insulin therapies cannot regulate glucose levels on a minute-to-minute level as the cells in the pancreas can. This, on the long term, might cause severe acute and long-term complications (including retinopathy, neuropathy, and nephropathy) which have significant social and economic impact. Transplantation of insulin producing cells is a proven option to prevent diabetic complications in a subset of patients with T1D. It requires lifelong immunosuppression which has side effects such reduced immune defense against pathogens. The treatment suffers from donor shortage.
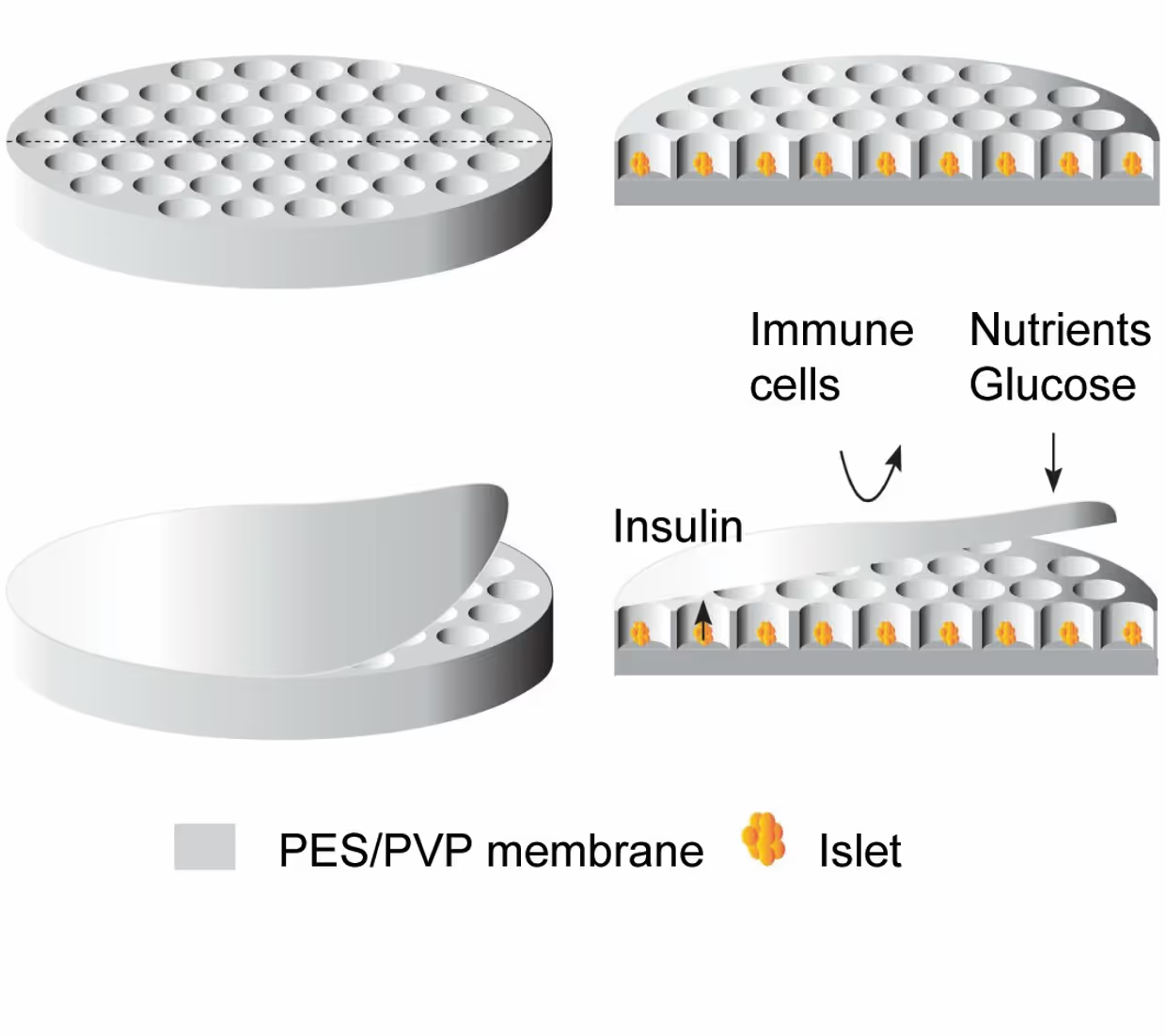
The (bio)artificial organs chair of Professor Dimitrios Stamatialis focuses on the development of novel immune-protective membrane-based cell encapsulation devices, containing insulin producing cells for regulation of blood glucose in T1D, without the need for injections and immunosuppressive drugs. The pancreatic islets are seeded in separate microwells to avoid aggregation, whereas the membrane porosity is tailored to achieve sufficient transport of nutrients, glucose and insulin (Figure 1).
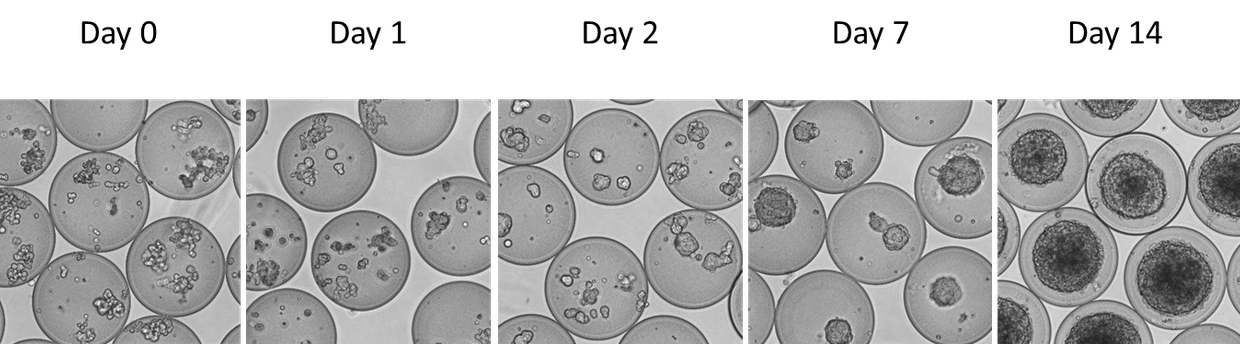
Within the Developmental BioEngineering group, professor Marcel Karperien and doctor Jeroen Leijten have developed microfabrication technologies for the encapsulation of insulin producing βcells in microcapsules consisting of enzymatically cross-linked polymers. These microcapsules have a diameter of approximately 150 micrometers, consist of a micrometer thin shell and a hollow core which contains the cells. The shell protects the cells from the host’s immune system while optimally facilitating diffusion of glucose and insulin. The cell containing microcapsules can be introduced in the patient using a simple injection (Figure 2).