Sustainable and highly stable ion exchange membranes for industrial applications
Hestie Brink (PhD Candidate), Wiebe de Vos (promotor), Saskia Lindhoud (co-promotor) |
Duration: 2022-2026 |
Funding: NWO - NWA, project HYPER |
Introduction
Electrochemical processes are becoming increasingly important as the world is striving to become less dependent on fossil fuels. Electrochemical cells are used in a wide range of applications and can either be used to convert chemical energy into electrical energy (batteries, fuels cells) or to drive non-spontaneous redox reactions by applying an external voltage (hydrogen generation from water). One of the main limitations of these processes is the poor performance and stability of the anion exchange membranes, especially at high salinities and extreme alkaline conditions.
Keywords
Anion exchange membranes, hot-pressing, saloplastics, polyelectrolyte complex, electrochemistry
Technological challenges
A novel approach to produce stable ion exchange membranes through hot-pressing of polyelectrolyte complexes was recently developed at the University of Twente. While these membranes showed great promise in terms of pH stability, electrical resistance and permselectivity, their ion exchange capacity and water uptake did not compare well with commercially available membranes.
Research goals
This research will focus on (i) improving the charge density and chemical stability of the hot-pressed anion exchange membranes and (ii) studying their performance in electrically driven processes.
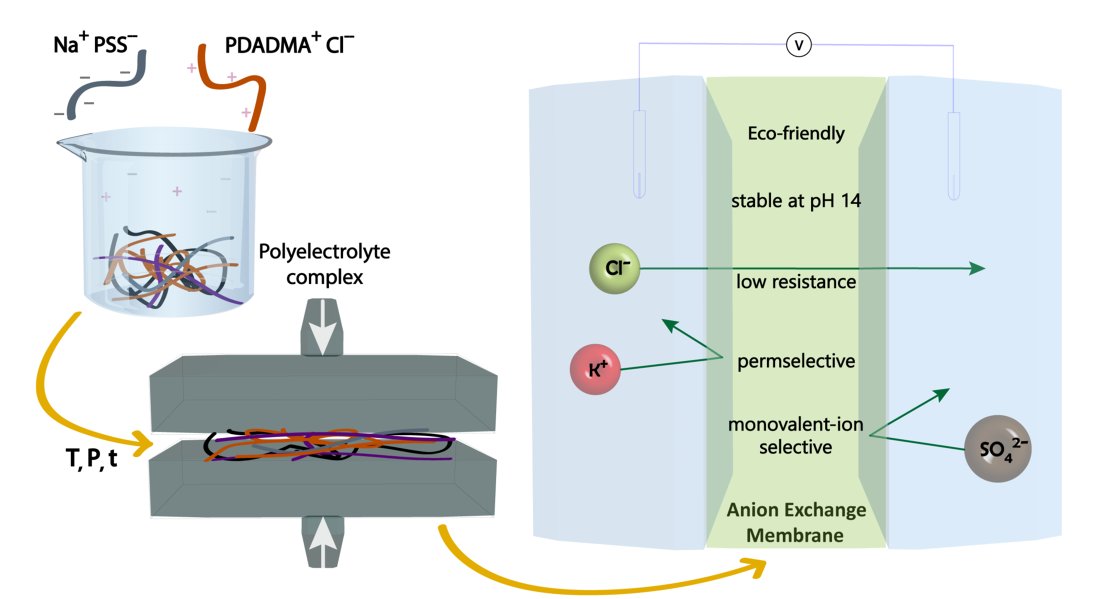
Picture derived from: Krishna, B.A., Lindhoud, S. and de Vos, W.M., 2021. Hot-pressed polyelectrolyte complexes as novel alkaline stable monovalent-ion selective anion exchange membranes. Journal of Colloid and Interface Science, 593, pp.11-20
