Tumour cells circulating in the blood of cancer patients are important markers for early diagnosis, treatment success and patient’s prognosis. How to recognise them, if there are just so few? Thanks to artificial intelligence, researchers of the University of Twente managed to automate the process with a high level of accuracy. And more than that. The new technology not only detects the tumour cells but also unravels hidden information. Extracellular vesicles, important in cell interaction, are classified as well. The researchers present their results in the February 10 issue of Nature Machine Intelligence.
Circulating Tumour Cells (CTCs) that are released from the original tumour and start moving in the blood stream, play an important role in the onset of metastases in other parts of the body. If you detect them in patient’s blood, this informs you about the effectiveness of treatment and about the patient’s prognoses. The number of CTCs is extremely small, so manually ‘counting’ them using fluorescence microscopy is labour intensive, even for an expert that knows how to recognise the CTCs. The new approach that is now presented, is a further step in the automation, with an accuracy of over 96 percent.
The new approach takes the existing one, for which UT scientists already developed an open-source recognition package that proved its clinical relevance, to a higher level. That is because it takes automated learning a decisive step further than the classic approach. Thanks to advanced visualisation, even more information comes out of a blood sample: CTCs of different types, as well as the extracellular ‘CD45 vesicles’ that give extra information on cell collaboration, for example. A lot of research is currently done on the role of these vesicles.
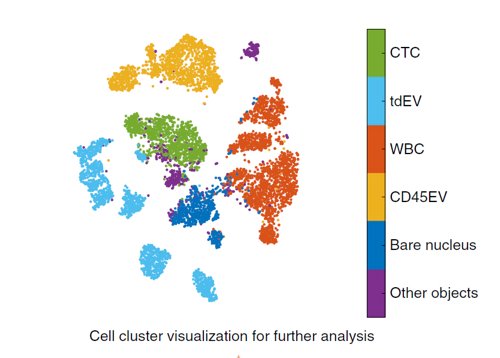
Map of cell types, CTCs (green), white blood cells (orange) and extracellular vesicles (yellow)
Window in black box
From deep learning networks we know that you train them with a large set of well-labelled examples, after which they will be able to learn and recognise by themselves, often even better than humans do. How the actual learning process takes place, what choices the network makes internally, is less transparent: although it’s successful, it is essentially a black box. Thanks to the extra modelling and visualisation step based on semi-supervised autoencoding, more information comes out of the network than expected. It is like you can see part of the learning through a small window in the black box. CTCs of different types, white blood cells and vesicles can be clearly distinguished.
The open-source image analysis programme called ACCEPT, developed by Leonie Zeune, can be combined with the CellSearch technology for detecting CTCs. This technology, co-invented by Prof Leon Terstappen, is already being used by some hospitals. Thanks to the deep learning development now presented, automated detection is further improved.
The paper ‘Deep learning of circulating tumour cells’, by Leonie Zeune, Yoeri Boink, Guus van Dalum, Afroditi Nanou, Sanne de Wit, Kiki Andree, Joost Swennenhuis, Stephan van Gils, Leon Terstappen and Christoph Brune, is published online at February 10 in Nature Machine Intelligence.





