Project aim and strategy
The main aim of this project is the fabrication of functional tissues, that can serve as next-generation implants and organ-on-chip devices, to mimic the healthy and damaged articular joint. The devices will be used for testing new therapeutic agents and may assist in reducing or replacing the use of experimental animals.
The main steps to achieve this goal will be:
- Development of novel bioinks;
- Integration of 3D printing and microfluidic technology to develop microfluidic devices for each tissue of the articular joint (e.g synovium, cartilage, bone);
- Replicating biochemical and biomechanical interactions of the joint as an organ in vitro.
Project partners
- Dr. Susana Piluso (Orthopaedics – UMCU & Developmental Bioengineering – UT)
- Dr. Liliana Moreira Teixeira Leijten (Developmental Bioengineering – UT & Equine Sciences – UU)
- Dr. Jeroen Leijten (Developmental Bioengineering – UT)
- Dr. Severine Le Gac (Applied Microfluidics for BioEngineering Research – UT)
- Prof. dr. Daniel Saris (Orthopaedics – UMCU, Developmental Bioengineering – UT)
- Prof. dr. Rene van Weeren (Equine Sciences – UU)
- Prof. dr. Marcel Karperien (Developmental Bioengineering – UT)
- Prof. dr. ir. Jos Malda (Orthopaedics – UMCU)
Highlights
The synovial joint is a complex organ comprising several tissues, such as articular cartilage, subchondral bone, synovium, menisci, tendon, ligament and fat pad. These tissues work closely together to maintain joint homeostasis, which is crucial for the proper functioning of the joint. The ideal in vitro model to capture this complex environment consist of several joint tissues-on-chip connected with each other in a modular fashion.
Highlight 1
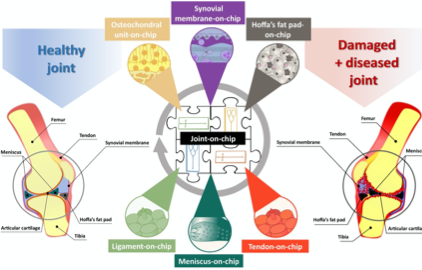
Osteoarthritis (OA) is a common invalidating disease characterized by gradual and unavoidable joint deterioration affecting 1.4 million Dutch. Current in vivo and in vitro models lack translation power for the development effective OA-treatments. We are currently developing an organ-on-chip model to mimic joint tissues typically affected by OA (e.g. cartilage, bone, synovial membrane). These on-chip units uniquely mimic the mechanical stimuli present in the joint, and with all units combined, will closely resemble a micro-joint.
Highlight 2
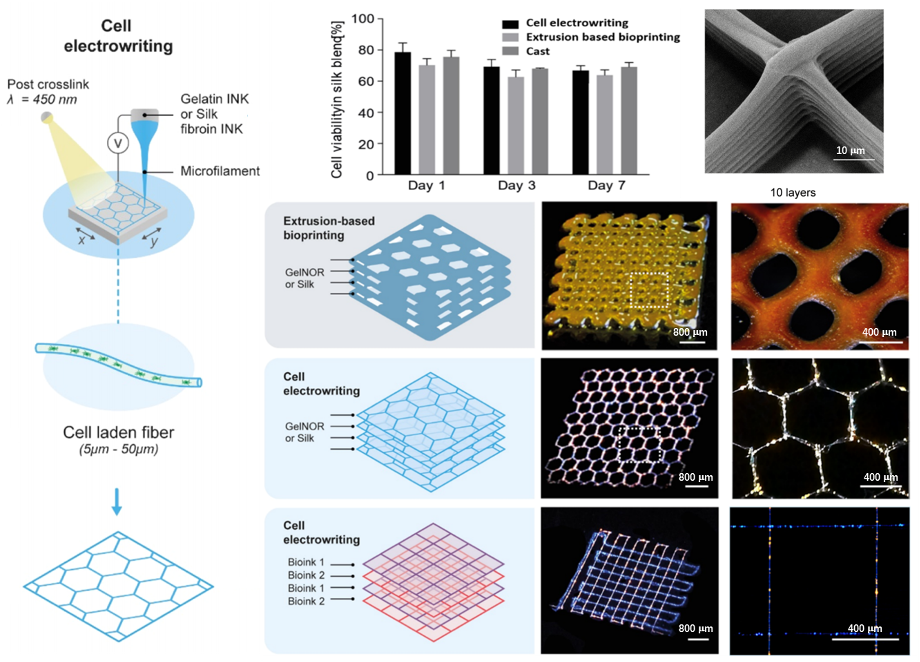
Biomacromolecules 2021, 22, 855−866
Silk fibroin, as a novel hydrogel-based bioink was combined with cell electrowriting to achieve well-organized cell-laden fiber structures with diameters ranging from 5 to 40 μm. More importantly, the biocompatibility of the bioink maintained a high cell viability post printing. Such unprecedented resolution and patterning precision open promising avenues for the high-resolution printing of labile biological moieties and living cells, leading to the creation of cellular microenvironments for regenerative medicine and organ-on-a-chip studies.
Highlight 3
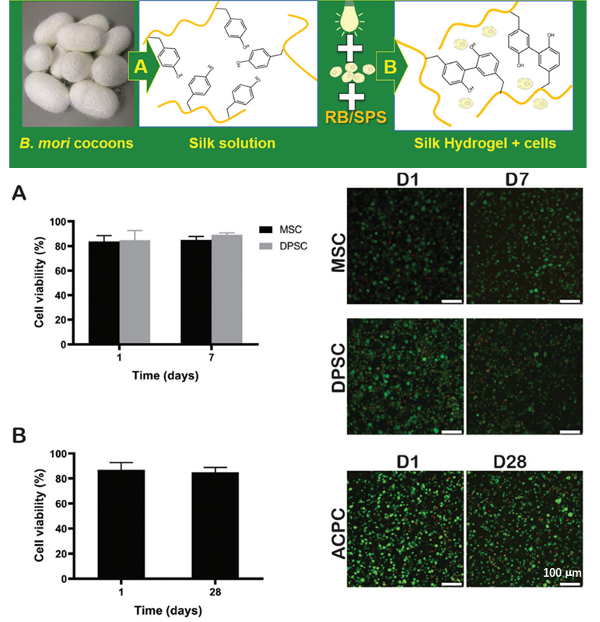
We conducted the first study in which cells were encapsulated within the rapid photo-crosslinking silk hydrogel matrix and were? present during the photo-chemical reaction. The silk hydrogel can support the viability of different mammalian cell types and could potentially act as a promising bioink formulation for bioprinting application.
Highlight 4

A bioinspired synovium-on-a-chip system to study synovial inflammation during osteoarthritis. Cyclic fluidic shear stress is applied to patient-derived synovial fibroblasts residing on silk hydrogel within the microfluidic chip, mimicking the redistribution of the synovial fluid during the extension and flexion of the knee joint. The release patterns of inflammatory biomarkers followed a similar pattern as in previous in vivo experiments, which renders the potential of such in vitro model for rapid screening of arthritic drugs.
Publications
- Piluso S, Li Y, Abinzano F, Levato R, Moreira Teixeira L, Karperien M, Leijten J, van Weeren R, Malda J. “Mimicking the Articular Joint with In Vitro Models”. Trends in Biotechnology. DOI: 10.1016/j.tibtech.2019.03.003
- Piluso, S., Flores Gomez, D., Dokter, I. , Moreira Texeira, L., Li, Y. , Leijten, J., Van Weeren, R., Vermonden, T. , Karperien, M., & Malda, J. (2020). Rapid and cytocompatible cell-laden silk hydrogel formation: Via riboflavin-mediated crosslinking. Journal of Materials Chemistry B, 8(41), 9566-9575. https://doi.org/10.1039/d0tb01731k
- Schwab A, Levato R, D'Este M, Piluso S, Eglin D, Malda J. Printability and Shape Fidelity of Bioinks in 3D Bioprinting. 2020, Chem Rev 120(19):11028-11055. doi: 10.1021/acs.chemrev.0c00084.
- Miguel Castilho, Riccardo Levato, Paulina Nunez Bernal, Mylène de Ruijter, Christina Y. Sheng, Joost van Duijn, Susanna Piluso, Keita Ito, and Jos Malda. Hydrogel-Based Bioinks for Cell Electrowriting of Well-Organized Living Structures with Micrometer-Scale Resolution. 2021, Biomacromolecules 22(2):855-866. DOI: 10.1021/acs.biomac.0c01577
- Kamperman, T., Teixeira, L. M., Salehi, S. S., Kerckhofs, G., Guyot, Y., Geven, M., Geris, L., Grijpma, D., Blanquer, S. & Leijten, J., Engineering 3D parallelized microfluidic droplet generators with equal flow profiles by computational fluid dynamics and stereolithographic printing. 7 Feb 2020, In: Lab on a chip. 20, 3, p. 490-495 6 p. https://doi.org/10.1039/c9lc00980a
- Baptista, D. , Teixeira, L. M. , Birgani, Z. T., van Riet, S. , Pasman, T. , Poot, A. , Stamatialis, D., Rottier, R. J., Hiemstra, P. S. , Habibović, P. , van Blitterswijk, C., Giselbrecht, S. , & Truckenmüller, R. (2021). 3D alveolar in vitro model based on epithelialized biomimetically curved culture membranes. Biomaterials, 266, [120436]. https://doi.org/10.1016/j.biomaterials.2020.120436
- Rodrigues, J. , Heinrich, M. A. , Teixeira, L. M. , & Prakash, J. (2021). 3D In Vitro Model (R)evolution: Unveiling Tumor–Stroma Interactions. Trends in cancer, 7(3), 249-264. https://doi.org/10.1016/j.trecan.2020.10.009
- Chou, D. B., Frismantas, V., Milton, Y., David, R., Pop-Damkov, P., Ferguson, D., MacDonald, A., Vargel Bölükbaşı, Ö., Joyce, C. E. , Moreira Teixeira, L. S., Rech, A., Jiang, A., Calamari, E., Jalili-Firoozinezhad, S., Furlong, B. A., O’Sullivan, L. R., Ng, C. F., Choe, Y., Marquez, S., ... Ingber, D. E. (2020). On-chip recapitulation of clinical bone marrow toxicities and patient-specific pathophysiology (Nature Biomedical Engineering, (2020), 4, 4, (394-406), 10.1038/s41551-019-0495-z). Nature Biomedical Engineering, 4(4), 477, https://doi.org/10.1038/s41551-019-0495-z, and Author correction https://doi.org/10.1038/s41551-020-0529-6
- Farzin, A. , Hassan, S. , Moreira Teixeira, L. S. , Gurian, M. , Crispim, J. F., Manhas, V., Carlier, A., Bae, H., Geris, L., Noshadi, I., Ryon Shin, S. , & Leijten, J. (2021). Self-Oxygenation of Tissues Orchestrates Full-Thickness Vascularization of Living Implants. Advanced functional materials, 31(42), [2100850]. https://doi.org/10.1002/adfm.202100850
- Moreira Teixeira Leijten, L. S., & Mezzanotte, L. (2021). New bioimaging avenues for organs-on-chips by integration of bioluminescence. View, 2(6), [20200177]. https://doi.org/10.1002/VIW.20200177
- Ahmed, H. M. M. A. M. , & Moreira Teixeira, L. S. (2022). New Endeavors of (Micro)Tissue Engineering: Cells Tissues Organs on-Chip and Communication Thereof. Cells tissues organs, 211(6). https://doi.org/10.1159/000516356
Acquired funding by PIs since start of program:
- ReumaNederland: Technovolution in Joint Repair (600k€, 2018-2022);
- ReumaNederland: Managing Joint Complexity (600k€, 2018-2022; LLP-12);
- ReumaNederland (600k€, 2018-2022; LLP-22).
- NWO Domain Science Open Competition ENW-KLEIN grant (news item)
- Incentive Grants for Women in STEM 2020-2022
- VIDI grant for Jeroen Leijten
- TTW Perspectief project “William Hunter Revisited’’
- TTW Perspectief project “SMART Organ-on-Chip: Standardized open Modular Approach to Recapitulate Tissue”
- LSH grant
Media
- Proefdiervrij gewrichtsonderzoek, doneren voor onderzoek via website Stichting Proefdiervrij, in Dutch
