BACKGROUND
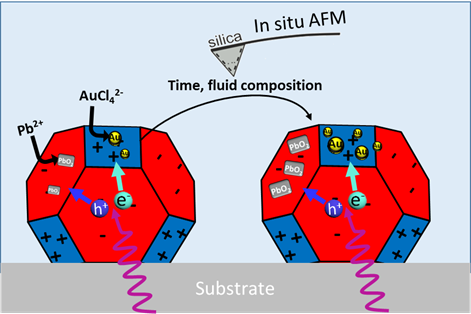
Direct semiconductor-based photocatalytic conversion of solar energy to chemical fuels is considered an ideal, renewable energy resource for the future. According to the current paradigm optimum performance is achieved by using faceted nanoparticles of semiconducting materials (SrTiO3 and BiVO4) and by functionalizing them in a facet-selective manner with cocatalysts (Pt, Au, PbO2 and MnOx) for the desired redox reactions, such as water splitting or CO2 reduction. Cocatalysts can provide trapping sites for the photogenerated charge carriers (as sinks) and extend the lifetime of these carriers. Also, cocatalysts can provide the active sites for the surface oxidation/reduction reaction through lowering the activation energy in multiple electrons transfer-involved hydrogen evolution reaction and oxygen evolution reaction. Among various methods used to load cocatalysts, photodeposition (i.e., photochemical deposition) has been considered as one of the most promising means, with the advantages of intimate contact (promoted charge transfer), easy preparation under a mild condition and site-directed loading. After photodeposition, traditionally, various ex situ characterizations (e.g., scanning electron microscopes) are performed to confirm the loading of cocatalysts and check their sizes/locations. However, the growth kinetics of cocatalysts during photodeposition is largely a black box. This lack of information partially leads to a relatively empirical optimization of cocatalysts during photodeposition in the field of photocatalysis to date. Here, we aim to use Dynamic Atomic Force Microscopy to image the photodeposition process of single cocatalysts on semiconductor faceted particles in situ. The Bachelor project will provide both novelty and challenges as it has never been applied to faceted SrTiO3/cocatalyst systems before.
RESEARCH OBJECTIVES
The goal is to directly study in situ the growth of individual cocatalyst nanoparticles on faceted particles of SrTiO3 and the rates at which they appear over time. To do this, high resolution Atomic Force Microscopy imaging in liquids and ultra-sharp tips will be used. We are particularly interested in how light intensity, wavelength, and fluid composition (pH and concentration of precursor ions AuCl4− and Pb2+) can affect the spatial distribution and growth kinetics of cocatalysts nanoparticles. These experiments can be complemented with SEM measurements.
LEARNING OBJECTIVES
In addition to the standard learning objectives for a Bachelor’s project (research planning, academic writing, data presenting, how to work in a lab environment, etc.), you will:
· Learn how to work with Atomic Force Microscope in air/liquids
· Acquire or increase your lab experience with colloidal semiconductor nanoparticles
· Learn fundamental concepts of photocatalysis and cocatalyst photo-deposition
CONTACT INFORMATION
