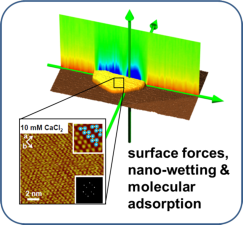
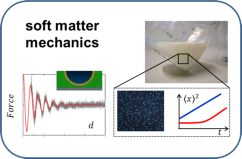
The research goal of PCF is to understand and to control liquids and their interfaces from molecular to macroscopic scales. Our research connects fundamental phenomena in static and dynamic wetting, nanofluidics, microfluidic two-phase flow, functional surfaces, chemical reactivity at interfaces to practically relevant applications. In recent years, our application focus has shifted towards challenges of the energy transition and CO2 mitigation, such as photocatalysis, mineral carbonation (olivine), biogas upgrading and advanced lubrication strategies, e.g. for electric vehicles and/or wind turbines.