2025
Mangut started his phd at Msus this week. We welcome him to the group!
2024
Congratulations to Roberto on winning the Mesa+ meeting picture prize!
Roberto won the picture prize at the MESA+ meeting this year for his SEM image titled "Trees of Particles on Crystal Soil". Congratulations on the prize, Roberto!

msus welcomes a new member to the group!
We extended a worm welcome to Ali Hameed this week who started as a PhD candidate!
Msus was present at euromembranes 2024 in prague!!!
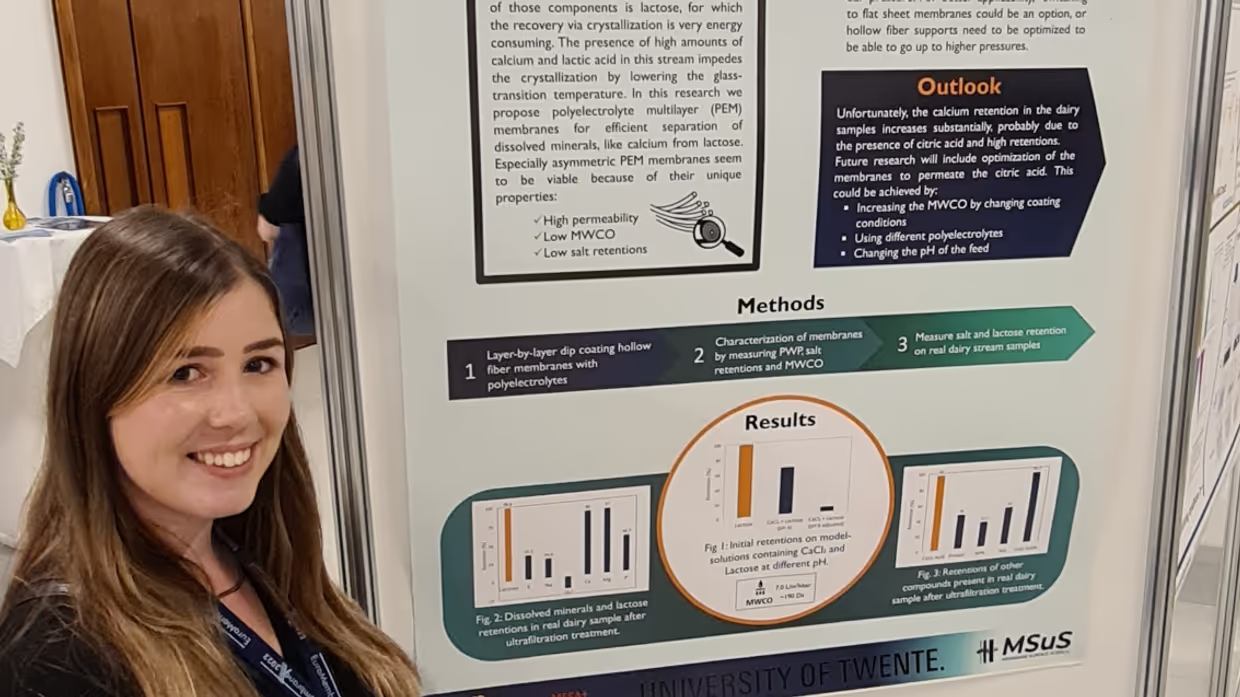
We shared our recent findings at Euromembrane this year! Xiao, Hestie, Halil, Tjerk, Esra and Wiebe all gave an oral lecture on use of polyelectrolytes as membranes! With talks varying from fundamental understanding of the polyelectrolytes to polyelectrolyte multilayer membranes for whey separation!
Wiebe de vos has been invited to speak at amk upcoming December
On December the 3rd Wiebe will give a talk about "Polyelectrolyte Based Nanofiltration membranes, Made to Order" at the Aachener Membran Kolloquium (AMK)

MSuS welcomes a new Collegue!
After performing her master thesis in our group Mia has decided to stay and join MSuS as a PhD candidate. She will be working on "Polyelectrolyte multilayer based membranes for solvent stable nanofiltration in Biorefinery"
M. Jurković MSc (Mia)
PhD Candidate
Jurjen Regenspurg published his recent work in desalination
Our colleagues shared their work at the Brightlands polymer days this year!
A delegation of our group was present at the Brightlands Polymer Days this May! Here Hestie Brink and Tjerk Watt gave talks about the recent developments in their work.

MSuS was present at the North American Membrane Society (NAMS) conference this year!
This year our colleague Tjerk Watt was present at the NAMS 2024 where he gave a talk about his recent work on "Improved nanofiltration through inverted membrane structures" By inverting the asymmetric porous structure of hollow fiber membranes he was able to coat defect free outer-skinned polyelectrolyte multilayer membrane (PEMM). Through this technique he was able to increase the active surface area of these PEMMs by a factor of 3.5 all while retaining the nanofiltration properties that make these polyelectrolyte multilayer type membranes ideal for the removal of organic micropollutants!

MSuS started the year by welcoming three new PhD candidates to the group!
2023
MSus wishes you a Happy hollidays and an exciting 2024!

MSUS attended ICOM 2023 last month!
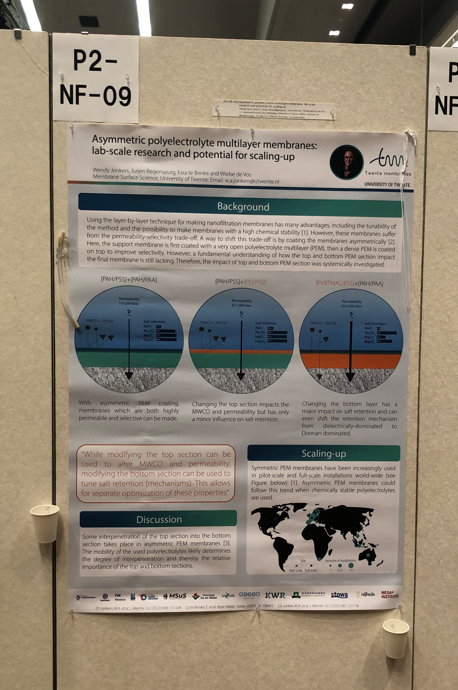
Last month, Wendy Jonkers presented her work on Asymmetric polyelectrolyte multilayer membranes at ICOM in Japan with a poster!
Today we held our annual msus group outing!
To celebrate the start of the summer, we held our annual group outing at Outdoor Holten this year! We had a day filled with fun activities and finished it off with a nice BBQ! There was a fierce competition as to who would be the best MSuS team, however, Wiebe proved to be no match for the rest of us and brought his team the victory on multiple events!
MSuS was present at the Dutch polymer days!
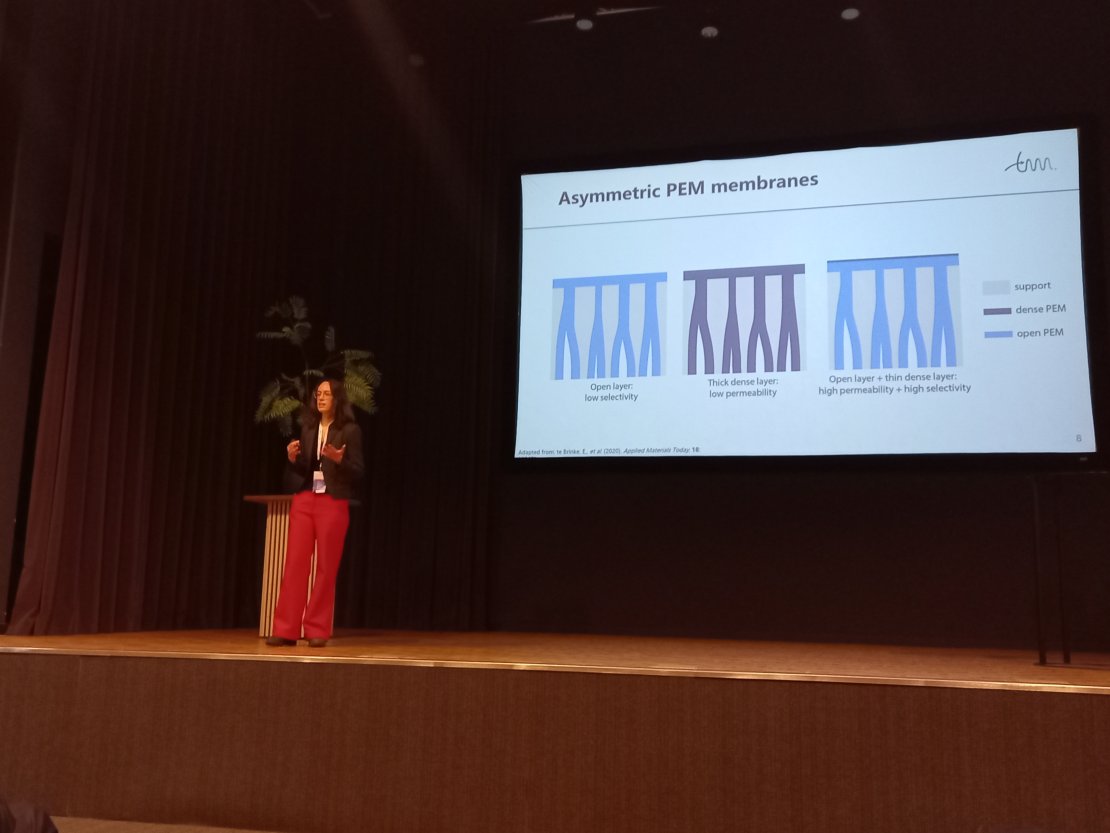
We presented our latest work at the Dutch Polymer Days this April! We were honoured to give multiple talks and a planery lecture by Wendy Jonkers about designing asymmetric polyelectrolyte multilayer membranes for efficient separations!
2 Million consolidator ERC grant awarded to Wiebe de vos for mosaic membranes!
Wiebe obtained a grant for 2 Million euros for his research project on mosaic membranes that are supposed to filter out micropollutants from wastewater while passing salts to avoid the formation of concentrated brines!
Wiebe: "I feel very lucky and honoured to have recieved an ERC Consolidator Grant. Together with my group, we will work on so-called charge-mosaic membranes, where alernating nanosized domains with negative and positive charges will bring unique separation properties."
More information on the project can be found in the article of University of Twente!

Tao Wang will defend his PHD thesis on the 9th of February!  Tao investigated catalytic membrane based advanced oxidation processes for micropollutant removal. In his thesis, catalytic membranes, which can successfully integrate membrane separation and sulfate radicals-based advanced oxidation processes (SR-AOPs) are developed. These membranes are then immediately applied to treat water streams containing organic micropollutants (MPs). Given the wide spread of MPs in our surface waters and their toxicities, MPs pose a significant threat, not only to aquatic organisms but also to human beings. Since wastewater treatment plants only provide limited removal to MPs, different technologies, including adsorption, membrane separation, and AOPs have been applied to remove MPs from wastewater. However, the diverse physicochemical properties of MPs make them very challenging when just a single technology is used. The integration of membrane separation and AOPs has the potential to provide sufficient removal efficiently. Indeed, the membrane is very suitable as a substrate for required small-sized catalysts, avoiding the difficulties of reusing and recycling them. Meanwhile, through activating the oxidants by the catalysts embedded within the membrane structure, the reactive species generated can degrade MPs when the solution passes through the membrane structure. In this case, not only the MP concentrations in the permeate are reduced but also the rejected MPs can be degraded, preventing the formation of the highly concentrated retentate. This thesis not only focuses on the fabrication of SR-AOPs-based catalytic membranes but also studies the influence of operational parameters on MPs removal.
Tao investigated catalytic membrane based advanced oxidation processes for micropollutant removal. In his thesis, catalytic membranes, which can successfully integrate membrane separation and sulfate radicals-based advanced oxidation processes (SR-AOPs) are developed. These membranes are then immediately applied to treat water streams containing organic micropollutants (MPs). Given the wide spread of MPs in our surface waters and their toxicities, MPs pose a significant threat, not only to aquatic organisms but also to human beings. Since wastewater treatment plants only provide limited removal to MPs, different technologies, including adsorption, membrane separation, and AOPs have been applied to remove MPs from wastewater. However, the diverse physicochemical properties of MPs make them very challenging when just a single technology is used. The integration of membrane separation and AOPs has the potential to provide sufficient removal efficiently. Indeed, the membrane is very suitable as a substrate for required small-sized catalysts, avoiding the difficulties of reusing and recycling them. Meanwhile, through activating the oxidants by the catalysts embedded within the membrane structure, the reactive species generated can degrade MPs when the solution passes through the membrane structure. In this case, not only the MP concentrations in the permeate are reduced but also the rejected MPs can be degraded, preventing the formation of the highly concentrated retentate. This thesis not only focuses on the fabrication of SR-AOPs-based catalytic membranes but also studies the influence of operational parameters on MPs removal.
For more information about his work you can visit this website
The defence will be streamed live through the following link starting from 12:30 on the 9th of February https://vimeo.com/event/2706159
2022
msus welcomes two guest phd candidates and a new phd candidate to the group!
msus was present at Euromembrane 2022 in sorrento
In November, MSuS was present at Euromembrane 2022. We enjoyed sharing our work through multiple talks and a poster presentation, and had many nice discussions about membranes. We hope to see everyone at the next edition!
MSUS welcomes three new phd candidates to the group!
ON THE 14TH OF JULY AMEYA KRISHNA BYSANI SUCCESSFULLY DEFENDED HIS PHD THESIS Hot-PRESSED SALOPLASTICS AS SUSTAINABLE ION-EXCHANGE MEMBRANES
Ameya Krishna B is a researcher in the Membrane Surface Science group led by Prof. Dr. ir. Wiebe M. de Vos (supervisor) and Dept. of Molecules and Materials supervised by Dr. Saskia Lindhoud (co-supervisor), Faculty of Science & Technology (S&T). In his work, complexes of charged polymers are softened with salt, and are used to fabricate dense material and further used as membranes for ion-exchange.

Further information about his work can be found here:
MSUS MASTERCHEFS- WENDY AND MATTHIJS, bag the pi-day bake-off prizes
JERE VAN LENTE OBTAINED HIS PH.D. ON THE 17TH OF MARCH
Jere's defense- APPLICATIONS OF POLYELECTROLYTE COMPLEXES FOR SEPARATION PROCESSES Jéré van Lente is a PhD student in the research group Membrane Science & Technology (MST). His supervisors are prof.dr.ir. W.M. de Vos and prof.dr. M.M.A.E. Claessens from the Faculty of Science & Technology (S&T).
 Polyelectrolytes are polymers with potentially charged monomer units. Under the right conditions, oppositely charged polyelectrolytes in solution can interact and form polyelectrolyte complexes (PECs). These PECs have peculiar emergent properties. One of these properties is the ability to accumulate certain molecules in very high concentrations. A comparison can be made between PECs and membraneless-organelles (MLOs), which are droplet-like liquid-in-liquid phases present in our cells that aid the cell in organising its interior milieu through selective partitioning of specific molecules. Similar to PECs, MLOs are also composed op oppositely charged polyelectrolytes. In the case of MLOs, these are typically negatively charged RNA molecules and positively charged intrinsically disordered proteins. Taking inspiration from nature, in this dissertation we present the use of PECs for the partitioning of molecules. We investigated whether it was possible to use PECs to selectively extract a protein from a mixture. Followed by whether a PEC containing a protein could be used as a biocatalytic membrane.
Polyelectrolytes are polymers with potentially charged monomer units. Under the right conditions, oppositely charged polyelectrolytes in solution can interact and form polyelectrolyte complexes (PECs). These PECs have peculiar emergent properties. One of these properties is the ability to accumulate certain molecules in very high concentrations. A comparison can be made between PECs and membraneless-organelles (MLOs), which are droplet-like liquid-in-liquid phases present in our cells that aid the cell in organising its interior milieu through selective partitioning of specific molecules. Similar to PECs, MLOs are also composed op oppositely charged polyelectrolytes. In the case of MLOs, these are typically negatively charged RNA molecules and positively charged intrinsically disordered proteins. Taking inspiration from nature, in this dissertation we present the use of PECs for the partitioning of molecules. We investigated whether it was possible to use PECs to selectively extract a protein from a mixture. Followed by whether a PEC containing a protein could be used as a biocatalytic membrane.
More information can be found at:
https://www.utwente.nl/en/education/tgs/currentcandidates/phd/calendar/2022/3/439311/phd-defence-jere-van-lente-applications-of-polyelectrolyte-complexes-for-separation-processes
2021
Jurjen's latest publication on Molecular Weight effects in PEM Nanofiltration Membranes
MSuS researchers present their work At euromembrane
Elif Nur durmaz defended her doctoral thesis successfully
The following accordion gives detailed information about her work:
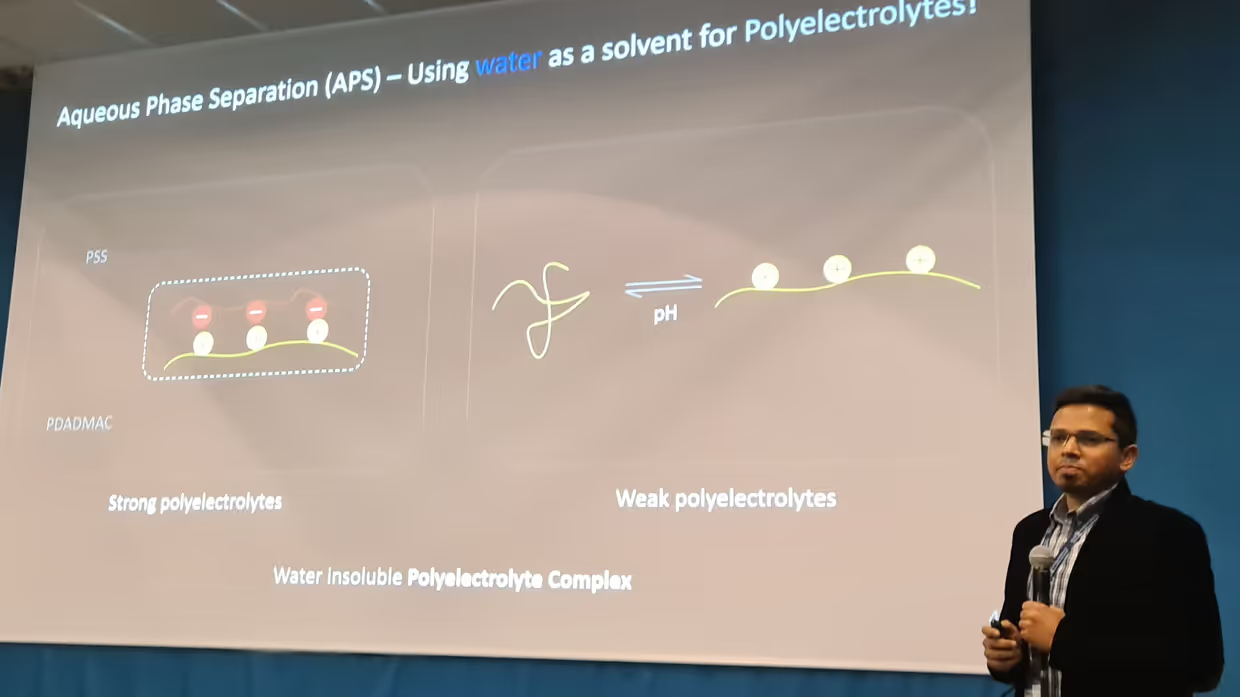
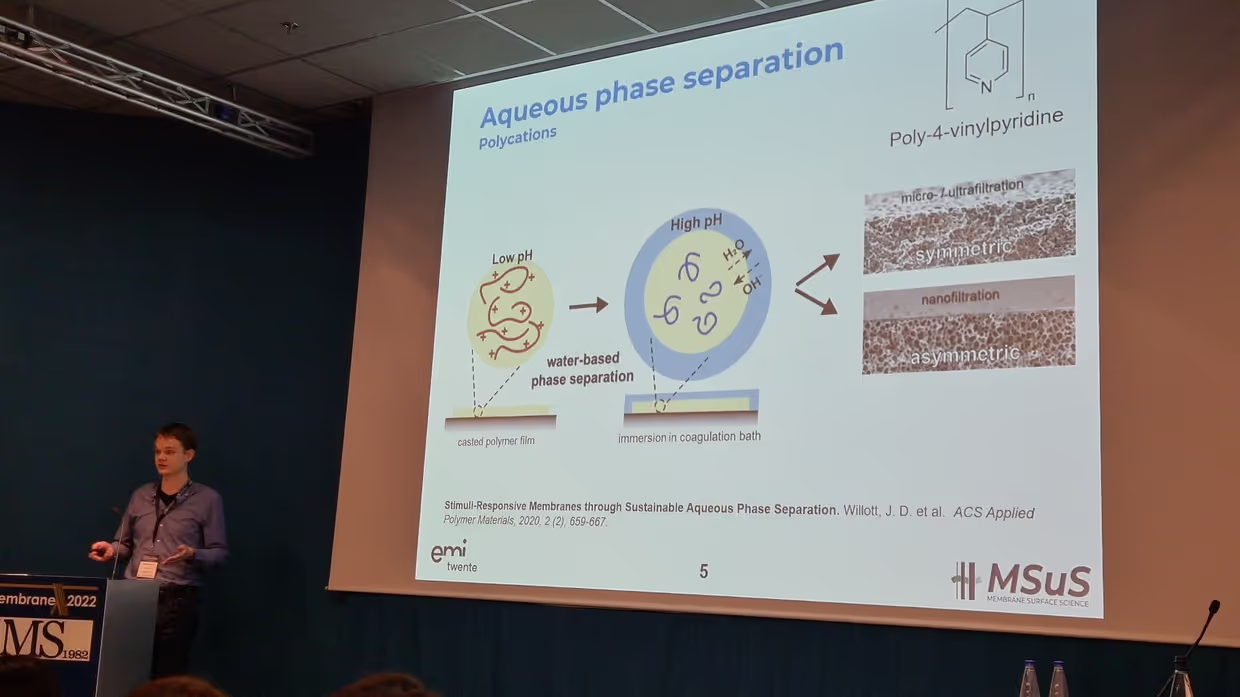
COMPLEX, INDEED: POLYELECTROLYTE-BASED MEMBRANES VIA AQUEOUS PHASE SEPARATION Elif's thesis focuses on membrane formation via polyelectrolyte complexation induced Aqueous Phase Separation (APS). Membranes are typically produced via phase separation methods in which toxic organic solvents are vital. The novelty of the APS approach is that only aqueous solutions are utilized to prepare membranes, which could eliminate the use of tons of organic solvents every year. Polyelectrolytes (PEs) are charged polymers and they have been used as membrane materials for decades, but mostly as membrane coatings or as dense films. Obtaining porous, free-standing membranes was not possible with conventional methods until APS methods came into the picture. Here, either the stimuli-responsiveness or the complex formation behavior of PEs can be used to achieve phase separation. There are two complexation-based APS approaches, namely pH-switch and salinity-switch. This thesis covers both of these approaches and a detailed discussion will take place on membrane formation and potential tuning parameters. In the first half of the thesis, the pH-switch approach is studied. Two different PE pairs were investigated. PE molecular weight, casting solution pH, coagulation bath pH, coagulation bath salinity, and PE mixing ratio are the tuning parameters that are affecting the membrane structure and performance.
With the pH-switch approach, membranes that can operate from microfiltration to nanofiltration applications were obtained. In the second half, poly(sodium 4-styrenesulfonate) (PSS), and poly(diallyldimethylammonium chloride) (PDADMAC) were studied for the salinity-switch approach. Mostly, nanofiltration type membranes were obtained. Coagulation bath salinity and PE mixing ratio are the parameters that effectively changed membrane performance. In the outlook chapter, possibilities for polyelectrolyte complex membranes including organic solvent filtrations and hollow fiber spinning are discussed. The thesis is concluded by highlighting that the know-how of the membrane formation with APS is achieved and that a critical point remains in the careful selection of the used materials.
A link to the details of her defense can be found below:
https://www.utwente.nl/en/education/tgs/currentcandidates/phd/calendar/2021/10/111326/phd-defence-elif-nur-durmaz-complex-indeed-polyelectrolyte-based-membranes-via-aqueous-phase-separation
IRSHAD defends his Ph.D. thesis and continues AT MSUS as a Postdoctoral researcher
Read on for details of his work and thesis:
SUSTAINABLE POLYELECTROLYTE COMPLEX MEMBRANES PRODUCED VIA AQUEOUS PHASE SEPARATION The following is a brief summary of his work and thesis:
In Chapter 1, we started this journey by discussing ‘sustainability’, especially in these crucial times when our relationship with the environment is strained. One way to repair our relationship with the environment is to develop, encourage and financially support sustainable manufacturing techniques. This can be achieved by implementing and utilizing the 12 principles of Green Chemistry as discussed in Chapter 1. Greener technologies and processes are the future and really the only way forward for the humankind and the Earth in the long run. One such effort to pursue a greener production of polymeric membranes has been presented in this thesis. We have successfully demonstrated that membranes can be prepared without using any unsustainable organic solvents. The polyelectrolyte complexation (PEC) based ‘Aqueous Phase Separation (APS)’ technique described in this thesis relies solely on water to produce polymeric membranes. This new technique provides a great deal of control over membrane structure and properties, just like the traditional organic-solvent based approaches. PEC membranes with tunable pore sizes have been prepared and optimized for a multitude of applications. The following sections summarize the major conclusions of the research work conducted in this thesis.
In Chapter 2 we explored polyelectrolyte complexation as one of the routes to obtain sustainable APS membranes. Here, the phase separation is achieved by changing the pH of the casting solution containing a strong polyanion i.e. poly(sodium 4-styrenesulfonate) (PSS) and a weak polycation i.e. poly(allylamine hydrochloride) (PAH). The casting solution is prepared by mixing the two polyelectrolytes in a monomer molar mixing ratio of 1:2 (PSS:PAH) at pH ~14. Immersing a thin film of this solution in a pH ~1 bath causes PAH to acquire charge and immediately form a polyelectrolyte complex with PSS. It was found that the molecular weights of the polyelectrolytes and the concentration of the casting solution are crucial parameters that have a significant impact on the membrane structure and properties. Utilizing polyelectrolytes having lower molecular weights typically resulted in porous PEC membranes that are well suited for microfiltration applications. The pore size of these membranes could be controlled from 0.2 µm down to 80 nm by simply varying the casting solution concentration, thereby affecting the kinetics of phase separation. On the other hand, utilizing higher molecular weight polyelectrolytes resulted in membranes having average pore sizes in the range of 2 – 5 nm, perfectly suited for tight ultrafiltration applications. Additionally, the kinetics of phase separation were controlled by adding NaCl to the precipitation bath, allowing the polyelectrolyte chains to rearrange into dense complexes. Nanofiltration type membranes, well suited for organic micropollutant (Mw 250 – 630 Da) removal, were obtained in this way. We successfully demonstrated that APS is indeed a versatile technique that provides great control over the membrane pore size and structure resulting in membranes that can be used for different applications. This greener approach to membrane fabrication completely eliminates the use of organic solvents while providing excellent membranes.
In Chapter 3, we explored various additional factors influencing the thermodynamics of solution and the kinetics of phase separation of the PSS-PAH based APS membranes. Since PAH is a weak polyelectrolyte, it exists in its charged state at neutral and acidic pH. Therefore, in order to prepare a casting solution, NaOH needs to be added to the PAH solution before mixing it with the PSS. The amount of added NaOH is critical as it determines the pH of the solution and as a result, the charge on PAH. A clear homogeneous solution cannot be obtained if the amount of added NaOH in PAH is not enough to completely remove its charge. However, adding significantly larger amounts of NaOH results in salting-out of PAH. Therefore, it is important to carefully balance the amount of NaOH added to PAH to obtain a clear PSS-PAH casting solution. We also find that the monomer mixing ratio of PSS:PAH significantly influences the mutual interactions of the polyelectrolytes in the casting solution thereby affecting the dynamic viscosity. Consequently, the kinetics of phase separation are severely impacted by the monomer mixing ratios resulting in membranes with different morphologies. Since this version of APS relies on the change in pH to induce phase separation, the pH of the coagulation bath becomes critically important since it is the driving force for complexation. It was found that if the driving force for complexation/phase separation is high, such as from solution pH of ~14 to coagulation bath pH ~0.5, the membranes formed instantaneously having dense and brittle structures. On the other hand, at higher bath pHs (pH > 1) the resultant membranes precipitated slower, had porous structures, and were mechanically weak. The pH of the coagulation bath, therefore, also controls the kinetics of polyelectrolyte complexation and the subsequent phase separation, and hence can be fine-tuned to obtain desired membrane morphology. Another important parameter that was found to affect the membrane structure and properties is the amount of crosslinking agent. Glutaraldehyde (GA) was added to the coagulation bath to crosslink the amine groups of PAH. It was revealed that the pore size of the membranes could be tuned from 2 nm through to 450 nm by varying the amount of GA in the coagulation bath. The stability of the PSS-PAH membranes was evaluated by immersing the membranes in highly saline solutions i.e. 1 M NaCl and 1 M KBr for 1 week. The results revealed that the morphology and performance of the membranes remain unaffected after exposure to saline solutions. Additionally, the membranes showed chemical stability against 1200 ppm sodium hypochlorite solution which is typically used as the cleaning agent for polymeric membranes. The results presented in Chapter 3 thus reveal the versatility of pH shift induced APS technique where numerous tuning parameters are available to produce PSS-PAH membranes with various structures, properties, and performances.
In Chapter 4, the applicability of the PSS-PAH membranes was taken one step further by showing that these membranes can also act as excellent support materials for functional coatings. Ultrafiltration type PSS-PAH membranes, obtained in Chapter 2, were coated with polyelectrolyte multilayers using three different polyelectrolyte pairs. It was found that only 4.5 bi-layers were required to transform the membranes into dense nanofiltration types that show a molecular weight cut-off (MWCO) in the range of 210 to 390 Da with salt retentions in excess of 90%. In addition, interfacial polymerization (IP) was performed on the PSS-PAH membranes to obtain nanofiltration membranes that yielded excellent separation performance. There are several benefits of using PSS-PAH membranes as supporting materials. For instance, the natural positive charge on the membranes facilitates the multilayer build up without any pre-treatments. Additionally, the excess amines of PAH provide additional bonding with the polyamide layer during IP, thereby improving the adhesion of the coating. These developments demonstrate that the APS approach allows membranes that can be used as excellent supports for functional coatings.
One major downside of these membranes described so far, is that they require extreme pH environments (from pH ~14 to pH ~1) to induce polyelectrolyte complexation. This problem was alleviated in Chapter 5 where polyethyleneimine (PEI) was used as a weak polyelectrolyte to prepare PSS-PEI membranes. The benefit of using PEI is that it is easily available in its uncharged state which means that a casting solution can directly be prepared by mixing PSS and PEI without adding any NaOH. Consequently, extreme pH, for example pH ~1, is not necessary to induce phase separation. The PSS-PEI solution can be precipitated at a benign pH of 4 to obtain PEC membranes. Similar to the PSS-PAH based APS membranes, the impact of several tuning parameters such as the molecular weight of PEI, the concentration and pH of the coagulation bath, and the amount of crosslinking agent was investigated in detail. Contrary to what was observed for the PSS-PAH membranes, using higher molecular weight polyelectrolytes resulted in microfiltration type PSS-PEI membranes and vice versa. This has to do with the branched nature of higher molecular weight PEI (~750 kDa) which forms a more open polyelectrolyte complex with PSS. On the other hand, excellent nanofiltration type membranes having a MWCO of ~200 Da were obtained using relatively lower molecular weight PEI (~ 25 kDa). PSS-PEI membranes performed even better as compared to the PSS-PAH membranes. In addition, the casting solution viscosity is significantly lower for the former at the same polymer concentration, making it easier to process for flat sheet membrane production. The major benefit, however, remains that these membranes can be prepared at mild pH conditions, making this APS approach even more sustainable and environmentally friendly.
In Chapter 6 an entirely different approach to prepare PEC membranes was taken where instead of pH, salt was used as the driving force for complexation. Here, a mixture solution of two oppositely charged strong polyelectrolytes i.e. PSS as polyanion and poly(diallyldimethylammonium chloride) (PDADMAC) as polycation, was prepared at high salt concentrations where the charges are screened. The solution is then cast as thin film and immersed in deionized water where the salt diffuses out of the solution leading to polyelectrolyte complexation and subsequent phase separation. Asymmetric nanofiltration membranes having MWCO of <300 Da with dense top layers and porous support were prepared via this approach. The effect of various parameters such as the molecular weights of the polyelectrolytes, the polymer solution concentration, and the bath salinity were investigated in detail. It was found that the salinity of the coagulation bath is a critical parameter that determines the rate of precipitation and the resulting thickness of the separation layer. The foremost advantage of this salt-induced APS is that it does not rely on changes in pH at all while producing sustainable nanofiltration type membranes.
In all described chapters above we described producing flat sheet sustainable membranes via the APS technique. An important step to further establish the versatility of APS lies in the production of hollow fiber (HF) membranes.
In Chapter 7 we showed that PSS-PEI based hollow fiber membranes can indeed be prepared via the APS technique. HF membranes for micro- and ultrafiltration applications were prepared by carefully tuning the fiber spinning conditions. This approach opens up a world of possibilities for APS membranes where also other polyelectrolyte systems such as the PSS-PAH and PSS-PDADMAC systems could be used for the production of excellent HF membranes. Additionally, efforts could also be devoted to studying other potential polyelectrolyte pairs to develop APS membranes for specific applications which will be discussed later in the coming sections.
Ettore virga graduates with a cum laude!

Details about his work and the defense can be found here:
Wouter Nielen successfully defends his PhD thesis
Wouter nielen successfully defends his PhD thesis A NOVEL SUSTAINABLE APPROACH FOR MEMBRANE PREPARATION
Wouter Nielen was a PhD student in the research group Membrane Science & Technology (MST). His supervisor is prof.dr.ir. W.M. de Vos from the Faculty of Science and Technology (S&T).
 His thesis investigates and demonstrates how polymeric membranes can be prepared using the new and more sustainable aqueous phase separation (APS) approach. Polymeric membranes are used on huge scales for kidney dialysis, wastewater treatment, drinking water production, and as a less energy intensive alternative to conventional industrial separation processes. However, the vast majority of polymeric membranes are produced via an unsustainable and environmentally unfriendly process which uses large amounts of reprotoxic chemicals like N‑methyl‑2‑pyrrolidinone (NMP) and dimethylformamide (DMF). Unlike the conventional methods the APS approach uses a water based system using a pH or salinity switch instead of toxic organic solvents to prepare membranes. This allows for a more sustainable membrane production process without the use of reprotoxic solvents. In the past few years three different APS approaches have been established; the first is the single-polyelectrolyte approach, where a responsive polyelectrolyte solution is precipitated by a pH switch. The second and third approach, on the other hand, use a two polyelectrolyte APS system where the complexation of oppositely charged polyelectrolytes forms the membrane either through a pH or a salinity switch. This thesis focusses on the single polyelectrolyte approach and investigates two different polyelectrolytes that can be used in this approach as well as various parameters that can be used to control and tune the membrane preparation.
His thesis investigates and demonstrates how polymeric membranes can be prepared using the new and more sustainable aqueous phase separation (APS) approach. Polymeric membranes are used on huge scales for kidney dialysis, wastewater treatment, drinking water production, and as a less energy intensive alternative to conventional industrial separation processes. However, the vast majority of polymeric membranes are produced via an unsustainable and environmentally unfriendly process which uses large amounts of reprotoxic chemicals like N‑methyl‑2‑pyrrolidinone (NMP) and dimethylformamide (DMF). Unlike the conventional methods the APS approach uses a water based system using a pH or salinity switch instead of toxic organic solvents to prepare membranes. This allows for a more sustainable membrane production process without the use of reprotoxic solvents. In the past few years three different APS approaches have been established; the first is the single-polyelectrolyte approach, where a responsive polyelectrolyte solution is precipitated by a pH switch. The second and third approach, on the other hand, use a two polyelectrolyte APS system where the complexation of oppositely charged polyelectrolytes forms the membrane either through a pH or a salinity switch. This thesis focusses on the single polyelectrolyte approach and investigates two different polyelectrolytes that can be used in this approach as well as various parameters that can be used to control and tune the membrane preparation.
In Chapter 1 an introduction to membrane technology and applications is provided along with an overview of parameters used in conventional polymeric membrane preparation, the non-solvent induced phase separation (NIPS) approach. Several alternative methods to prepare polymeric membranes, which use different and less toxic solvents than the conventional NIPS method, are discussed. These methods aim to improve the sustainability of polymeric membrane production but ultimately still rely on organic solvents which the APS approach tries to avoid.
The preparation of membranes using the pH-responsive polymer poly(4‑vinyl pyridine) (P4VP) is discussed in Chapter 2. P4VP is a polybase and is dissolved at low pH where the polymer is charged and water-soluble, and subsequently casting the solution as a thin film. Switching to a high pH where the polymer becomes uncharged and insoluble results in controlled phase separation and solidification of the polymer into porous membrane structures. By tuning the pH of the coagulation bath and the polymer and acid concentration in the polymer casting solution a large control over the membrane structure is achieved. It was found that by using acetic acid instead of hydrochloric acid in the polymer casting solution the highest control over structure and reproducibility was achieved, most likely due to the weak acid acting as a buffer. By using these tuning parameters both symmetric porous microfiltration membranes and asymmetric dense nanofiltration membranes were prepared. In addition, the pH-responsive nature of P4VP automatically leads to a pH-responsive membrane where the degree of responsive behaviour can be tuned by the degree of crosslinking. This responsive behaviour makes it possible to easily clean the membrane, without using harsh chemicals.
While good permeability and retentions were achieved with the P4VP membranes, the mechanical stability was found to be suboptimal, as failure of the membranes was observed at an applied pressure of more than 4 bar. To prepare membranes with high mechanical stability using the APS approach the copolymer polystyrene-alt-maleic acid (PSaMA) was used in Chapter 3. PSaMA combines the responsive monomer maleic acid, required for APS, with the unresponsive hydrophobic styrene which enhances the mechanical stability of the resultant membranes. The precipitation of PSaMA can be precisely controlled which allows the preparation of a wide range of membranes, from microfiltration membranes capable of treating oily waste water, to dense nanofiltration-type membrane with excellent micropollutant retentions. As with the P4VP system weak acids again are a key parameter in controlling the precipitation behavior of the polyelectrolyte. Acetic acid is used in the polymer casting solution, where it brings the polymer close to the point of precipitation, as well as in the coagulation bath where it controls the precipitation rate of PSaMA. By tuning the final pH of the coagulation bath MF and UF membranes are prepared and by using phosphoric acid instead of acetic acid in the coagulation bath dense NF membranes. The mechanical stability of the PSaMA membranes is significantly better than the P4VP membrane prepared in Chapter 2 as the membranes presented in this chapter demonstrate stable operation up to 20 bar of applied pressure.
The phase behavior of polyelectrolytes can be determined by the pH and salinity, but also by the exact nature of the used counter-ions. In Chapter 4 it is investigated how salt type can affect, and thus be used to control, the phase separation of PSaMA. At equal ionic strengths, it is found that Na2SO4 and LiCl lead to the formation of more open structures compared to NaCl, NaNO3, NH4Cl, and MgCl2, while CaCl2 leads to densification of the top layer. These ion-specific effects appear to be caused by a combination of ion mobility and the interaction potential between the ion and the polymer. With increasing salt concentration similar effects are observed for all salt types, the formation of asymmetric membranes with denser top layers. Overall, it is shown that salt identity and concentration are key parameters in the APS process, and can be used to prepare both open microfiltration as well as dense nanofiltration membranes.
While in Chapter 3 and 4 good membranes have been prepared, large amounts of acetic or phosphoric acid were used in the coagulation baths. To further improve the sustainability of the APS approach it is important to reduce the amounts of acid used and prepare membranes in milder conditions. In Chapter 5 it is demonstrated that it is possible to prepare membranes with PSaMA while only using low concentrations (0.1–0.3 M) of hydrochloric acid in the coagulation bath. Additionally, it is shown that polymer and acetic acid concentration in the polymer casting solution have significant effects on the membrane formation. Polymer concentration in APS behaves the same as in regular NIPS, where an increased concentration leads to denser structures and vice versa. In Chapter 5 this is employed to prevent defect formation in the selective layer of the nanofiltration membranes. Differences in the acetic acid concentration in the polymer casting solution appear to mostly affect the support structure of the membrane where a reduced concentration significantly reduces the formation of macrovoids. Understanding of how these parameters affect membrane preparation and performance is essential to optimizing membranes prepared with APS.
The membranes prepared with PSaMA are typically chemically cross-linked to prevent the polymer from redissolving at neutral pH conditions. As crosslinking is a common method to make membrane resistant to organic solvents it is of interest to investigate if the standard crosslinking of PSaMA membranes also makes them solvent resistant. In Chapter 6, it is demonstrated that PSaMA membranes have stable performance in both isopropanol (IPA) and toluene, and a slightly reduced performance in N-methyl-2-pyrollidone (NMP). However, PSaMA does not perform well as a selective layer in these solvents, indicating that the real opportunity would be to use the UF type PSaMA membranes as solvent stable support membranes. Additionally, in Chapter 6 the pH stability and responsiveness of PSaMA membranes is also investigated. The membranes are shown to be stable in neutral and acid pH regimes (2–7) and the NF membranes demonstrate a pH dependent retention of Mg2+ and SO42− ions while the UF type membranes show a strong pH responsive behavior in regards to permeability. However, long term exposure to elevated pH conditions (pH 8–10), result in severe swelling of the NF membranes resulting in defect formation, and compaction of the UF membranes. For the UF membranes this compaction was found to be reversible for some but not all of the membrane samples measured. These results show that in aqueous systems membranes prepared with PSaMA have interesting responsive behavior, but perform best at neutral and acidic pH values. Moreover, the membranes exhibited excellent stability in the organic solvents IPA and toluene.
While the single-polyelectrolyte APS approach is extensively investigated in Chapter 2–6 research is never finished and there is plenty of work left to be done. In Chapter 7 an overview of different avenues to continue the investigation into the single-polyelectrolyte APS approach is provided. This includes a selection of interesting polymers specifically designed for the APS approach which can lead to new and improved membranes. Another important step in development of APS is the preparation of hollow fiber membranes, which offers some distinct advantages over flat sheet membranes, such as reduced fouling and higher packing densities. Initial results are shown which demonstrate that it is possible to prepare hollow fibers using the single-polyelectrolyte APS approach. Additionally, some issues that were encountered concerning the polymer quality are discussed which can have a severe impact on the formation on (defect free) membranes.
Overall, this thesis showcases the single-polyelectrolyte APS approach, a highly novel sustainable approach to prepare polymeric membranes. The approach offers distinct advantages over conventional membrane preparation methods as it does not rely on reprotoxic solvent. Moreover, it matches the versatility of the conventional methods as it allows for the preparation of both open and dense membranes. The prepared membranes can match commercial available membranes with retention in aqueous environments, have high mechanical stability and are resistant to various organic solvents, while especially the permeability still requires improvement. This thesis marks the beginning of a new method that has the potential to significantly improve the sustainability of the membrane industry.
Elif reviews polyelectrolyte membranes with advanced functionalities in her latest publication
Jiaying's fresh take on sustainable packaging using polyelectrolyte complexation as oxygen barrier coatings:
Want to know a fun way to make transparent saloplastics and use them as a membrane? Check out Ameya's paper:
2020
Ettore Virga wins the Marcel Mulder award 2020. MSUS is proud!

Ettore's research is part of the Concentrates Theme at Wetsus. He has invented a new generation of membranes that can prevent fouling even in the most adverse conditions. A novel zwitterionic polymer has been successfully coated on membranes to make them less prone to fouling. In this way, the industry will use less energy and chemical consumption for water filtration. The coating can be applied to any surface where fouling needs to be avoided, creating an even bigger impact.
Ettore Virga will graduate within the standard 4 years and already has published 6 papers on his new technology, all in high ranking journals. He is now in charge of coordinating the Concentrates Theme while finishing his Ph.D. research.
in her paper, Elif DURMAZ explains how she used SALINITY CHANGE to induce AQUEOUS PHASE SEPARATION, and in turn fabricate novel Polyelectrolyte Complex Membranes!
TÜRKAN'S LATEST ARTICLE ON 'DEFECT-FREE HOLLOW FIBER REVERSE OSMOSIS MEMBRANES' IS UP FOR VIEWING:
Anna Marie's new review on the 'Recent Developments and Practical Feasibility of Polymer‐Based Antifouling Coatings' is online!
Irshad bags the first prize at NAMS 2020 for his poster!
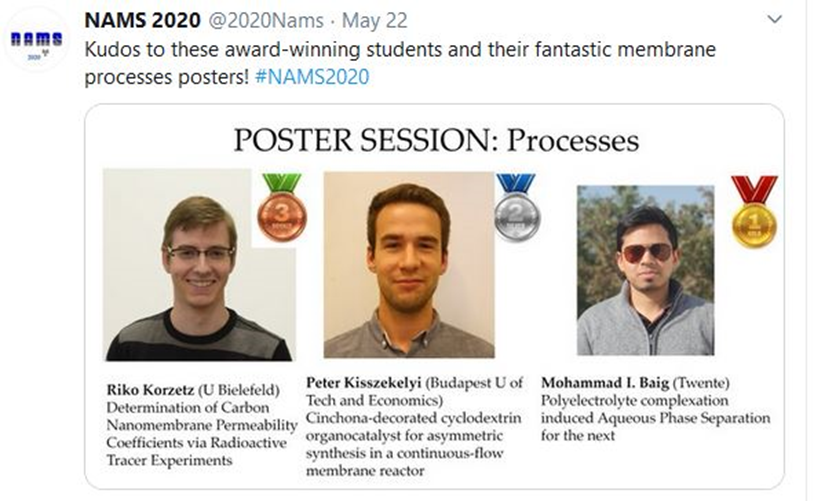
Muhammad Irshad Baig presented a poster titled “Polyelectrolyte complexation induced Aqueous Phase Separation for the next generation of sustainable membranes” at the North American Membrane Society (NAMS 2020) conference for which he received the Best Poster Award in the ‘Processes’ category. In this poster, Irshad highlights a new approach to prepare Aqueous Phase Separation membranes using a combination of strong and weak polyelectrolytes.
HOT OFF THE PRESS! Check out Wouter Nielen's latest article
Wouter's work on 'Aqueous Phase Separation of Responsive Copolymers for Sustainable and Mechanically Stable Membranes' is up for viewing!
2019
the LATEST article by Josh and Wouter is online!
Irshaid Baig's New publication in Advanced Functional materials!
And that's how we celebrated EsRa's Last day at work
PUBLICATION NEWS: ESRA's new paper!
Ameya Bysani wins 2nd prize of the David reinhoudt poster award!
News off the press! new Papers by Ettore Virga, Janneke Dickhout and Zamidi Ahmad
MSuS Day OUT! Activities, Lots of fun, and great food :)
5 new masters in Membrane Engineering! 
The MST cluster is proud to announce that on the 12th of July, 5 students successfully defended their MSc thesis. These students are all part of the Erasmus Mundus Master in Membrane Engineering for a Sustainable World (EM3E-4SW), a European MSc program run by 6 universities from 5 European countries. Three of the five students performed their final MSc project within the MSuS group. We congratulate Micah, Fei, Fatima, Jomuel, and Sajjad with their degree and wish them well. In the picture, the five students are with their supervisors.
Dr. Wiebe de Vos has featured in the crew of eye-openers! Eye-openers is an online stage for talented young scientists. Here they tell about their research, what's it about, what inspires them and what they're dreaming of. In clear language, visually stunning and in one minute only. Click the link below to watch what Wiebe has to say:
MSuS heartily welcomes three new Ph.D. candidates Jurjen, Lily, and Tao to the group!
2018
PhD defense of Jannecke Dickhout UNDERSTANDING MEMBRANE FOULING IN PRODUCED WATER TREATMENT
Janneke Dickhout was a PhD student in the Soft Matter, Fluidics and Interfaces Group. Her supervisor was prof.dr. R.G.H. Lammertink from the Faculty of Science and Technology.
 Produced water (PW) is the largest waste stream from the petrochemical industry (about 3 barrels of PW per barrel of oil). PW is an oil-in-water emulsion containing dispersed and dissolved hydrocarbons, surface-active compounds, solid particles and usually has a high salinity. This complex mixture has to be treated before the water can be disposed or re-used, and membrane treatment is a viable method to achieve this. Membranes, however, suffer from fouling, but the extent to which this results from the many different components in PW and/or from interactions between these many components is poorly understood. This is worrying, as understanding of the causes and mechanisms of membrane fouling, is essential to develop the membrane materials and membrane processes that would allow successful PW treatment.
Produced water (PW) is the largest waste stream from the petrochemical industry (about 3 barrels of PW per barrel of oil). PW is an oil-in-water emulsion containing dispersed and dissolved hydrocarbons, surface-active compounds, solid particles and usually has a high salinity. This complex mixture has to be treated before the water can be disposed or re-used, and membrane treatment is a viable method to achieve this. Membranes, however, suffer from fouling, but the extent to which this results from the many different components in PW and/or from interactions between these many components is poorly understood. This is worrying, as understanding of the causes and mechanisms of membrane fouling, is essential to develop the membrane materials and membrane processes that would allow successful PW treatment.
The aim of this thesis is to create increased understanding regarding membrane fouling by oil-in-water emulsions. The adhesion of oil droplets in an emulsion to a model surface was studied visually in a flow cell and compared to membrane filtration experiments using the same emulsion. It was found that the interaction between the salt concentration and surfactant type of the emulsion plays an important role in membrane fouling. Understanding this interaction is key to work towards new and improved membrane applications in challenging feed streams, such as PW.
PHD defense of Terica Sinclair 
Terica Sinclair, who worked on "Virus Control by Enhanced Physical Separation", received her Doctorate on the 31st of May, 2018. She was a Ph.D. student in MSuS, in collaboration with Wetsus, and was supervised by prof.dr.ir. H.D.W. Roesink and prof.dr. A.M. de Roda Husman.
Her research involved the modification of MF membranes for enhanced pathogen removal for drinking water treatment. By using these modified membranes, drinking water purification processes can be simplified compared to UF based treatment systems, a significant advantage for decentralized water treatment systems.
She received the 'Best Ph.D. project' award in June 2017. Details regarding her Ph.D. research can be found here.
PhD defense of Timon Rijnaarts 
Timon Rijnaarts successfully defended his Ph.D. on the 3rd of May, 2018. His study which was titled "The role of membranes in the use of natural salinity gradients for Reverse Electrodialysis" was in the MSuS group, in collaboration with Wetsus in Leeuwarden. His research focused on addressing challenges in Reverse Electrodialysis with natural waters. He studied different chemistries and shapes of polymeric membranes and alternative process strategies for Electrodialysis.
Since March 2018, Timon is working as a researcher for the European Membrane Institute (EMI), on water-related research.




























 Tao investigated catalytic membrane based advanced oxidation processes for micropollutant removal. In his thesis, catalytic membranes, which can successfully integrate membrane separation and sulfate radicals-based advanced oxidation processes (SR-AOPs) are developed. These membranes are then immediately applied to treat water streams containing organic micropollutants (MPs). Given the wide spread of MPs in our surface waters and their toxicities, MPs pose a significant threat, not only to aquatic organisms but also to human beings. Since wastewater treatment plants only provide limited removal to MPs, different technologies, including adsorption, membrane separation, and AOPs have been applied to remove MPs from wastewater. However, the diverse physicochemical properties of MPs make them very challenging when just a single technology is used. The integration of membrane separation and AOPs has the potential to provide sufficient removal efficiently. Indeed, the membrane is very suitable as a substrate for required small-sized catalysts, avoiding the difficulties of reusing and recycling them. Meanwhile, through activating the oxidants by the catalysts embedded within the membrane structure, the reactive species generated can degrade MPs when the solution passes through the membrane structure. In this case, not only the MP concentrations in the permeate are reduced but also the rejected MPs can be degraded, preventing the formation of the highly concentrated retentate. This thesis not only focuses on the fabrication of SR-AOPs-based catalytic membranes but also studies the influence of operational parameters on MPs removal.
Tao investigated catalytic membrane based advanced oxidation processes for micropollutant removal. In his thesis, catalytic membranes, which can successfully integrate membrane separation and sulfate radicals-based advanced oxidation processes (SR-AOPs) are developed. These membranes are then immediately applied to treat water streams containing organic micropollutants (MPs). Given the wide spread of MPs in our surface waters and their toxicities, MPs pose a significant threat, not only to aquatic organisms but also to human beings. Since wastewater treatment plants only provide limited removal to MPs, different technologies, including adsorption, membrane separation, and AOPs have been applied to remove MPs from wastewater. However, the diverse physicochemical properties of MPs make them very challenging when just a single technology is used. The integration of membrane separation and AOPs has the potential to provide sufficient removal efficiently. Indeed, the membrane is very suitable as a substrate for required small-sized catalysts, avoiding the difficulties of reusing and recycling them. Meanwhile, through activating the oxidants by the catalysts embedded within the membrane structure, the reactive species generated can degrade MPs when the solution passes through the membrane structure. In this case, not only the MP concentrations in the permeate are reduced but also the rejected MPs can be degraded, preventing the formation of the highly concentrated retentate. This thesis not only focuses on the fabrication of SR-AOPs-based catalytic membranes but also studies the influence of operational parameters on MPs removal.










 Polyelectrolytes are polymers with potentially charged monomer units. Under the right conditions, oppositely charged polyelectrolytes in solution can interact and form polyelectrolyte complexes (PECs). These PECs have peculiar emergent properties. One of these properties is the ability to accumulate certain molecules in very high concentrations. A comparison can be made between PECs and membraneless-organelles (MLOs), which are droplet-like liquid-in-liquid phases present in our cells that aid the cell in organising its interior milieu through selective partitioning of specific molecules. Similar to PECs, MLOs are also composed op oppositely charged polyelectrolytes. In the case of MLOs, these are typically negatively charged RNA molecules and positively charged intrinsically disordered proteins. Taking inspiration from nature, in this dissertation we present the use of PECs for the partitioning of molecules. We investigated whether it was possible to use PECs to selectively extract a protein from a mixture. Followed by whether a PEC containing a protein could be used as a biocatalytic membrane.
Polyelectrolytes are polymers with potentially charged monomer units. Under the right conditions, oppositely charged polyelectrolytes in solution can interact and form polyelectrolyte complexes (PECs). These PECs have peculiar emergent properties. One of these properties is the ability to accumulate certain molecules in very high concentrations. A comparison can be made between PECs and membraneless-organelles (MLOs), which are droplet-like liquid-in-liquid phases present in our cells that aid the cell in organising its interior milieu through selective partitioning of specific molecules. Similar to PECs, MLOs are also composed op oppositely charged polyelectrolytes. In the case of MLOs, these are typically negatively charged RNA molecules and positively charged intrinsically disordered proteins. Taking inspiration from nature, in this dissertation we present the use of PECs for the partitioning of molecules. We investigated whether it was possible to use PECs to selectively extract a protein from a mixture. Followed by whether a PEC containing a protein could be used as a biocatalytic membrane.




 His thesis investigates and demonstrates how polymeric membranes can be prepared using the new and more sustainable aqueous phase separation (APS) approach. Polymeric membranes are used on huge scales for kidney dialysis, wastewater treatment, drinking water production, and as a less energy intensive alternative to conventional industrial separation processes. However, the vast majority of polymeric membranes are produced via an unsustainable and environmentally unfriendly process which uses large amounts of reprotoxic chemicals like N‑methyl‑2‑pyrrolidinone (NMP) and dimethylformamide (DMF). Unlike the conventional methods the APS approach uses a water based system using a pH or salinity switch instead of toxic organic solvents to prepare membranes. This allows for a more sustainable membrane production process without the use of reprotoxic solvents. In the past few years three different APS approaches have been established; the first is the single-polyelectrolyte approach, where a responsive polyelectrolyte solution is precipitated by a pH switch. The second and third approach, on the other hand, use a two polyelectrolyte APS system where the complexation of oppositely charged polyelectrolytes forms the membrane either through a pH or a salinity switch. This thesis focusses on the single polyelectrolyte approach and investigates two different polyelectrolytes that can be used in this approach as well as various parameters that can be used to control and tune the membrane preparation.
His thesis investigates and demonstrates how polymeric membranes can be prepared using the new and more sustainable aqueous phase separation (APS) approach. Polymeric membranes are used on huge scales for kidney dialysis, wastewater treatment, drinking water production, and as a less energy intensive alternative to conventional industrial separation processes. However, the vast majority of polymeric membranes are produced via an unsustainable and environmentally unfriendly process which uses large amounts of reprotoxic chemicals like N‑methyl‑2‑pyrrolidinone (NMP) and dimethylformamide (DMF). Unlike the conventional methods the APS approach uses a water based system using a pH or salinity switch instead of toxic organic solvents to prepare membranes. This allows for a more sustainable membrane production process without the use of reprotoxic solvents. In the past few years three different APS approaches have been established; the first is the single-polyelectrolyte approach, where a responsive polyelectrolyte solution is precipitated by a pH switch. The second and third approach, on the other hand, use a two polyelectrolyte APS system where the complexation of oppositely charged polyelectrolytes forms the membrane either through a pH or a salinity switch. This thesis focusses on the single polyelectrolyte approach and investigates two different polyelectrolytes that can be used in this approach as well as various parameters that can be used to control and tune the membrane preparation.









 Produced water (PW) is the largest waste stream from the petrochemical industry (about 3 barrels of PW per barrel of oil). PW is an oil-in-water emulsion containing dispersed and dissolved hydrocarbons, surface-active compounds, solid particles and usually has a high salinity. This complex mixture has to be treated before the water can be disposed or re-used, and membrane treatment is a viable method to achieve this. Membranes, however, suffer from fouling, but the extent to which this results from the many different components in PW and/or from interactions between these many components is poorly understood. This is worrying, as understanding of the causes and mechanisms of membrane fouling, is essential to develop the membrane materials and membrane processes that would allow successful PW treatment.
Produced water (PW) is the largest waste stream from the petrochemical industry (about 3 barrels of PW per barrel of oil). PW is an oil-in-water emulsion containing dispersed and dissolved hydrocarbons, surface-active compounds, solid particles and usually has a high salinity. This complex mixture has to be treated before the water can be disposed or re-used, and membrane treatment is a viable method to achieve this. Membranes, however, suffer from fouling, but the extent to which this results from the many different components in PW and/or from interactions between these many components is poorly understood. This is worrying, as understanding of the causes and mechanisms of membrane fouling, is essential to develop the membrane materials and membrane processes that would allow successful PW treatment.
