HERE WE PROVIDE SHORT OVERVIEWS OF RESEARCH LINES WITHIN THE MSUS GROUP
INTERACTIONS AT THE MEMBRANE INTERFACE
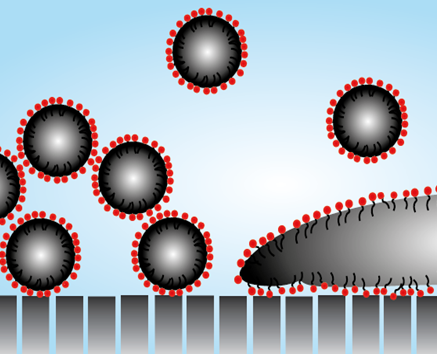
Fig. 1 Schematic picture of well stabilized (left) and poorly stabilized (right) oil droplets at the interface of an ultrafiltration membrane.
In complex feed mixtures such as surface waters, biological systems, and industrial wastewaters it can be very difficult to understand and predict membrane performance. A key example is produced water, that contains oil droplets, surfactants, salts, inorganic particles and dissolved organics (e.g. benzene, toluene). Membrane Technology is a very promising technique for the treatment (cleaning) of such produced water, but fouling and a lack of oil rejection are common problems. The complexity of such systems can be overwhelming, but by using well-characterized feed streams and careful experiments it does become possible to understand the major effects playing a role. For example, well-stabilized oil droplets will form an open cake layer (figure 1, left side) while poorly stabilized droplets will form an oil layer blocking the porous structure (figure 1, right side). Clearly, the oil droplets are the main fouling agent, but the chemistry of the water (surfactants, salts, pH) determines the fouling severity (Dickhout et al. 2017, JCIS, 487, 523-534).
MEMBRANE COATINGS
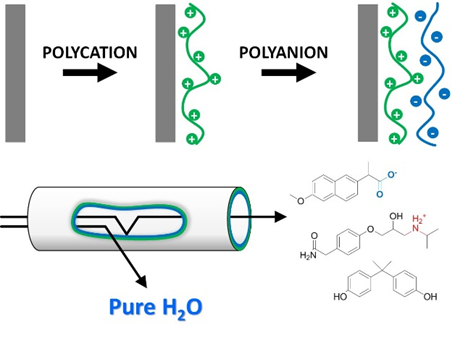
Figure 2. A polyelectrolyte multilayer on the inside of a hollow fiber UF membrane imbues it with NF/RO type separation properties to remove small organic molecules.
A coating can imbue a membrane with new separation properties and can allow an ultrafiltration membrane to become a nanofiltration membrane (Grooth et al. 2015, JMS, 475, 311-319), or allow a microfiltration membrane to remove viruses (https://www.utoday.nl/science/64543/safe-drinking-water-for-developing-countries). But within MSuS, we always strive to create coatings that are multifunctional, for example, coatings that provide the desired separation properties while at the same time leading to anti-fouling, responsive and/or easy to clean membranes (Ilyas et al. JCIS, 446, 386-39). Another important driver for the group is the development of novel NF/RO type membranes specifically designed for the removal of so-called micro-pollutants (figure 2). These are small organic molecules (100-1000 Dalton) that stem from industrial, medicinal and agricultural waste, and that can damage the environment and human health already at very low concentrations. For these coatings, we apply self-assembly of oppositely charged polyelectrolytes at the interface of a porous support membrane. In this so-called Layer-by-Layer assembly, the support membrane is alternatively exposed to polycations and polyanions, to build polyelectrolyte multilayer’s (PEMs) of controllable thickness. A large advantage of this approach is that the properties of the PEM layer, responsible for the separation properties of the membrane, can be tuned by choice of polymer and by the employed coating conditions such as pH and ionic strength. This method thus allows the design of a membrane truly optimized towards micro-pollutant removal, while, for example, still allowing small ions to permeate (Ilyas et al. 2016, JMS, 537, 220-228)
ADVANCED MEMBRANE MATERIALS
Under the right conditions, membrane formation and membrane functionalization can be attained in a single step. For example, the use of diblock-copolymer self-assembly for membrane fabrication not only leads to very uniform pore sizes (figure 3a), but membranes can be made responsive to pH and temperature by choice of the right block-copolymer. Control over the exact composition of the solvent during membrane casting provides further control over the membrane structure (Vriezekolk et al. JMS, 2016, 504, 230-239). But controlled precipitation of responsive polymers is also a highly promising method to create novel membranes completely from water. In the following figure (3), we show an example of a porous membrane prepared completely under aqueous condition. The membrane retains a responsive nature. Clearly organic solvents are not always required to obtain promising membrane structures.

Fig 3. SEM image of a porous membrane prepared under aqueous conditions
FUNDAMENTALS OF POLYMER AT SOLID-WATER INTERFACES
Coating solid surfaces with water-soluble polymers can be used to control various macroscopic phenomena including lubricating, adhesive and anti-microbial properties. To understand these ‘life-size’ behaviours it is first vital to understand the nanoscale properties of such layers. In many cases the behaviour (thickness, charge, solvation, structure) of the polymer changes in response to environmental conditions like pH, salinity, salt type and temperature. A part of what we do in MSuS is perform and compare experiments with computer modeling (numerical self-consistent modeling) and vice versa to gain fundamental insight into the behaviour of polymers at the solid-liquid interface (e.g. Willott et al. 2016, Macromolecules 49, 9605-9617).
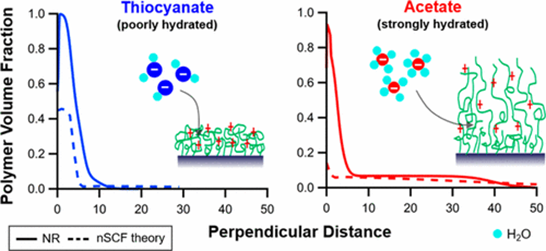
Fig 4. Comparison of neutron reflection experiments with nSCF theory for the response of a weak polyelectrolyte brush to different types of salt anions.
For more information, feel free to contact w.m.devos@utwente.nl.
