The second generation photoacoustic 3D tomographic breast imager
PROJECT ACRONYM PAM2
FUNDED BY Health∼Holland LSI-TKH PPP project SACAMIR (No. LSHM17007), and Stichting Achmea Gezondheidszorg for funding in Project No. Z620.
PRINCIPAL RESEARCHER Sjoukje Schoustra (M3I & BMPI) present affiliation Ecare
SUPPORT Johan van Hespen
SUPERVISORS Srirang Manohar (M3I) & Wiendelt Steenbergen (BMPI)
COLLABORATION PA Imaging R&D BV, Medisch Spectrum Twente (MST)
CLINICAL BACKGROUND [1] Breast cancer is the most frequently diagnosed cancer as well as the leading cause of cancer death among women worldwide [2]. Imaging is an important aid in the detection, diagnosis, treatment monitoring and follow-up of breast cancer [3]. Conventional imaging techniques currently used in the clinic are x-ray mammography (MMG), ultrasonography (US) and magnetic resonance imaging (MRI) [4]. MMG is the only method utilized in organized screening programs in several countries around the globe, since it is the only method proven to reduce mortality [5]. It does, however, require (often painful) breast compression, makes use of ionizing radiation and the detection sensitivity in dense breasts is relatively low [6]. US is used only in the diagnostic trajectory, and is often employed for targeted examinations of palpable lesions and lesions detected in screening. It is also the primary method used for guiding biopsies. It does not use ionizing radiation, but has the drawback of being operator dependent [7]. MRI is used in the diagnostic trajectory, often as a problem-solving modality. It does not use ionizing radiation, has high resolutions, and possesses high sensitivity. However, it requires the administration of contrast agents, does not have a high specificity, is expensive and not widely disseminated [4, 8]. Each technique has its strengths but also its weaknesses, leaving room for new imaging techniques that can potentially overcome the current limitations and drawbacks.
OBJECTIVE The goal of the thesis work was the development, testing, optimization and clinical demonstration of the 2nd Generation instrument the Photoacoustic Mammoscope 2 (Figure 1) developed in collaboration with the UT start-up, PA Imaging BV.  Figure 1: The PAM 2 system with (a) the bed on which a woman lies prone, (b) a foot rest, and (c) the breast aperture with (d) a schematic of the shape and position of the imaging tank below the bed, (e) the power supply and cooling unit, (f) laser head (behind the panel), (g) DAQ unit, and (h) step stool. (From Schoustra et al (2019) J. Biomed Opt., 24(12), 121909)
Figure 1: The PAM 2 system with (a) the bed on which a woman lies prone, (b) a foot rest, and (c) the breast aperture with (d) a schematic of the shape and position of the imaging tank below the bed, (e) the power supply and cooling unit, (f) laser head (behind the panel), (g) DAQ unit, and (h) step stool. (From Schoustra et al (2019) J. Biomed Opt., 24(12), 121909)
PROJECT DETAILS [1]
System Characterization The PAM 2 system was characterized and its performance assessed through measurements on test objects and healthy volunteers. The tomographic system made use of two illumination wavelengths (755 and 1064 nm), illuminating the breast (pendant in an imaging tank filled with water) from the nipple side and through multiple bundles directed at parts of the breast close to the chest wall. Twelve arc-shaped ultrasound detection arrays, placed in the imaging tank and each containing 32 piezocomposite elements, detected generated ultrasound waves from around the breast. A filtered backprojection algorithm reconstructed acquired data into 3D images. Absorbing beads on threads were imaged and reconstructed with a resolution of 1 mm along all axes. Detailed 3D images of vasculature in the human healthy breast (Figure 2) were acquired with the system. The need for immobilizing and positioning the breast during a scan of a few minutes arose over the course of this work.
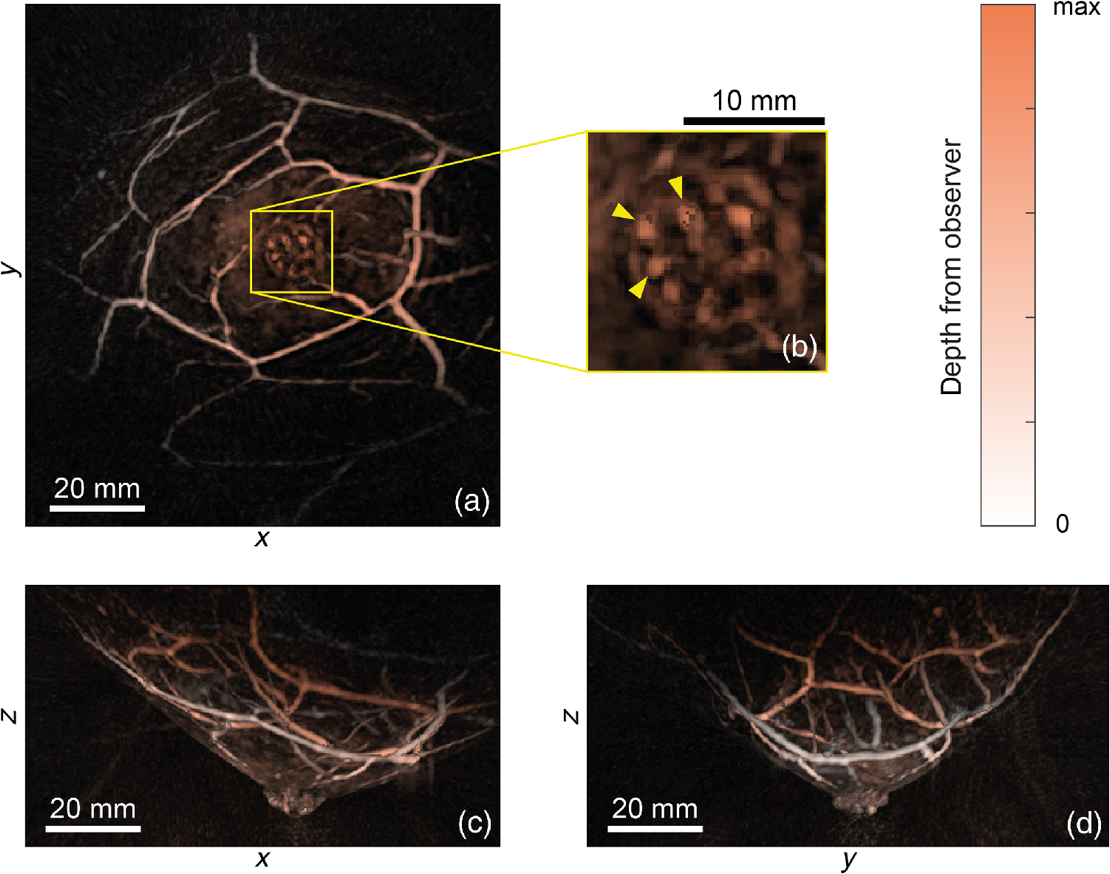 Figure 2 Color-coded images of the left breast of a healthy volunteer, acquired using PAM2, illuminated with 755 nm in three orientations (a) coronal, (c) sagittal, and (d) transverse. The zoomed subsection (b) shows the nipple features. (From Schoustra et al (2019) J. Biomed Opt., 24(12), 121909
Figure 2 Color-coded images of the left breast of a healthy volunteer, acquired using PAM2, illuminated with 755 nm in three orientations (a) coronal, (c) sagittal, and (d) transverse. The zoomed subsection (b) shows the nipple features. (From Schoustra et al (2019) J. Biomed Opt., 24(12), 121909
Breast-supporting cups The next step in optimization was the design, development and added value of breast-supporting cups to immobilize and position the breast. The design of the cup is rotationally symmetric: a two-dimensional (2D) curve (consisting of an ellipse and a cubic polynomial, the latter providing a rounded edge) is revolved to obtain a 3D structure. The cups were vacuum shaped from a sheet of polyvinyl chloride (PVC), a material tested to have a high optical transmission and acceptable acoustic transmission losses. Holes were punched in the cups, to let water from the imaging tank inside the cup during a measurement. Eight different sizes of cups were designed and tested, where healthy subjects’ breasts were scanned with multiple sizes cups and without support. The unsupported pendant breast was found to undergo a slow settling movement during the first two minutes. Moreover, breathing movement patterns were observed. Movements with use of a supporting cup were limited to ~0.1 mm.
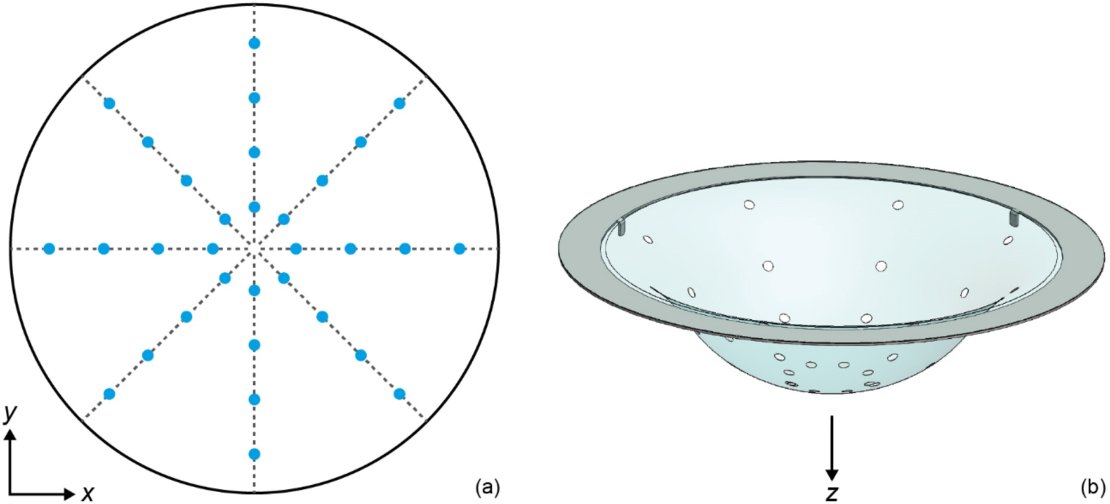
Figure 3 (a) Top-view sketch of a cup and the pattern of 32 holes (5 mm diameter) for water coupling. (b) 3D rendering of a cup with holes, mounted on a stainless steel ring. (From Schoustra et al (2021) Photoacoustics, 21, 100238)
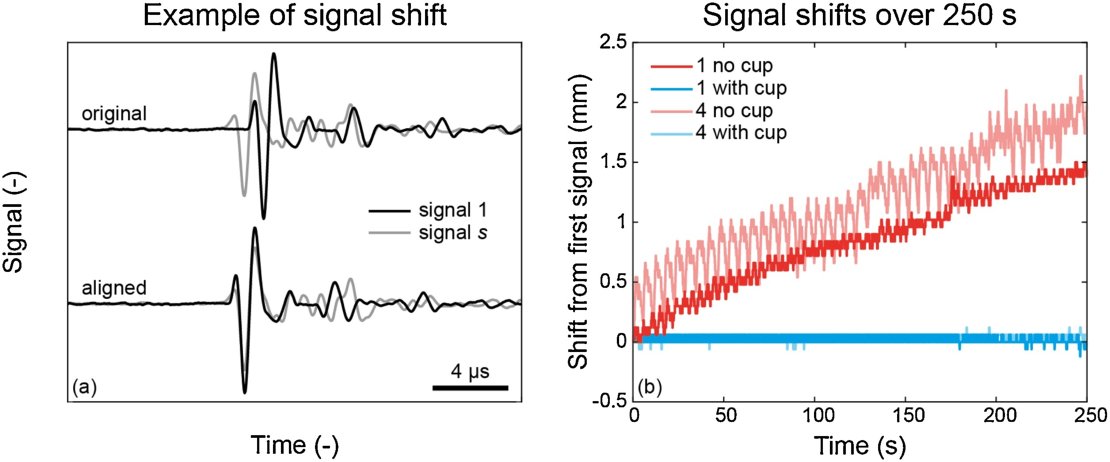 Figure 4. (a) Example of two signals before and after alignment. (b) Shift required for alignment of all 2500 signals measured by one detecting element, acquired over a time period of 250 s, with respect to the first acquired signal in that measurement. (From Schoustra et al (2021) Photoacoustics, 21, 100238)
Figure 4. (a) Example of two signals before and after alignment. (b) Shift required for alignment of all 2500 signals measured by one detecting element, acquired over a time period of 250 s, with respect to the first acquired signal in that measurement. (From Schoustra et al (2021) Photoacoustics, 21, 100238)
In this work, the filtered backprojection reconstruction introduced earlier was supplemented with an iterative algorithm, to enhance image quality. Delineation of the water-tissue boundary eased the implementation of a 2-speed of sound (2-SOS) model for image reconstruction, employing an SOS for the breast tissue inside the cup and a different SOS for the surrounding water. This enhanced image quality further. Overall, the use of cups to support and position the breast provided higher quality images, compared to images of the same breast measured without support. This was achieved by reducing movement during the measurement and by enabling the use of a more detailed reconstruction through a 2-SOS model. Moreover, the cups ensured central positioning of the breast within the imaged volume. Multiple sizes of the cups accommodated a range of breast sizes.
Clinical Study This work included clinical measurements on 30 breast cancer patients. Obtained photoacoustic images were compared to conventional clinical images: x-ray, ultrasound, and if available, magnetic resonance (MR) imaging. Patients presenting with a suspicious lesion were invited to participate in the study and undergo PAM 2 scans in between or directly after regular examinations. Of thirty three included patients, thirty were actually measured, of which nineteen were finally diagnosed with one or more malignancies, which we focused on in this work. A selection of four patients was made to study in detail. Reconstructed photoacoustic images (using an iterative reconstruction algorithm) were processed to enhance visibility of vascular structures and decrease background noise. Three of four selected patients also received dynamic contrast-enhanced (DCE) MR imaging, which was of use in locating the tumoral region by finding similar vascular structures in photoacoustic and MR images. After locating the tumoral region in the photoacoustic images, we searched for markers indicative of malignancy in this region. These results will be posted after publication of the article.
Steps towards longitudinal photoacoustic imaging This work describes the first steps towards longitudinal photoacoustic imaging, investigating the influence of the menstrual cycle on photoacoustic breast appearance. A first step was made to assess PAM 2 system repeatability with phantom measurements. Five phantoms with varying absorption and scattering characteristics were created and scanned multiple times. Image metrics based on contrast were extracted from all scans, and statistically analyzed. Intraclass correlation coefficients of calculated contrast values in scans on the five phantoms indicated good to excellent system repeatability. Next to phantom measurements, the breast of one healthy subject was scanned multiple times, where positioning variations between scans were apparent, hampering accurate longitudinal assessment. Recommendations were given for future research on this topic. These results will be posted after publication of the article.
OPPORTUNITES
We need highly creative, industrious and passionate students to help us move the field further. The backgrounds required are in Biomedical Engineering, Applied Physics, Technical Medicine, and Computer Science. If you are interested in making an important contribution to this area, contact the following:
PUBLICATIONS
- Schoustra, S.M., Piras, D., Huijink, R., Op't Root, T.J., Alink, L., Kobold, W.M.F., Steenbergen, W. and Manohar, S. (2019) Twente Photoacoustic Mammoscope 2: system overview and three-dimensional vascular network images in healthy breasts. Journal of Biomedical Optics, 24(12), p.121909. https://doi.org/10.1016/j.pacs.2020.100238
- Schoustra, Sjoukje M., Tim JPM Op't Root, Rutger P. Pompe van Meerdervoort, Laurens Alink, Wiendelt Steenbergen, and Srirang Manohar. (2021) "Pendant breast immobilization and positioning in photoacoustic tomographic imaging." Photoacoustics 21, 100238. https://doi.org/10.1117/1.JBO.24.12.121909
- Schoustra, Sjoukje M., Roeland Huijink, Laurens Alink, Tim JPM op't Root, Daan Sprünken, Daniele Piras, Wouter F. Muller Kobold et al. "The Twente photoacoustic mammoscope 2: 3D vascular network visualization." In Photons Plus Ultrasound: Imaging and Sensing 2019, vol. 10878, pp. 63-69. SPIE, 2019. https://doi.org/10.1117/12.2507448
- S. M. Schoustra, B. De Santi, T. J. P. M. op ‘t Root, R. P. Pompe van Meerdervoort, L. Alink, C. A. H. Klazen, M. C. van der Schaaf, J. Veltman, W. Steenbergen, S. Manohar (2022) Imaging breast malignancies with the Twente Photoacoustic Mammoscope 2 Under review
- S. M. Schoustra, B. J. Voorthuis, J. Veltman, W. Steenbergen, S. Manohar (2022) Towards longitudinal photoacoustic imaging of the healthy breast to investigate influence of the menstrual cycle to be submitted
References
- S. M. Schoustra “A photoacoustic 3D tomographic breast imager bench & bedside experiences. PhD Thesis, University of Twente. (Date of defence 16 September 2022)
- L. A. Torre et al., “Global cancer statistics, 2012,” CA Cancer J. Clin., 65 (2), 87 –108 (2015). https://doi.org/10.3322/caac.21262
- P. A. T. Baltzer et al., “New diagnostic tools for breast cancer,” Mag. Eur. Med. Oncol., 10 (3), 175 –180 (2017). https://doi.org/10.1007/s12254-017-0341-5
- L. W. Bassett, S. Lee-Felker, “Breast imaging screening and diagnosis,” The Breast, 337361.e2 5th ed.Elsevier(2018).
- D. A. Berry et al., “Effect of screening and adjuvant therapy on mortality from breast cancer,” N. Engl. J. Med., 353 (17), 1784 –1792 (2005). https://doi.org/10.1056/NEJMoa050518
- D. S. Al Mousa et al., “What effect does mammographic breast density have on lesion detection in digital mammography?,” Clin. Radiol., 69 (4), 333 –341 (2014). https://doi.org/10.1016/j.crad.2013.11.014
- R. J. Hooley, L. M. Scoutt and L. E. Philpotts, “Breast ultrasonography: state of the art,” Radiology, 268 (3), 642 –659 (2013). https://doi.org/10.1148/radiol.13121606
- B. E. Adrada, R. Candelaria and G. M. Rauch, “MRI for the staging and evaluation of response to therapy in breast cancer,” Top. Magn. Reson. Imaging, 26 (5), 211 –218 (2017). https://doi.org/10.1097/RMR.0000000000000147

