Noninvasive blood analysis in newborns
Bring me to the available projects!
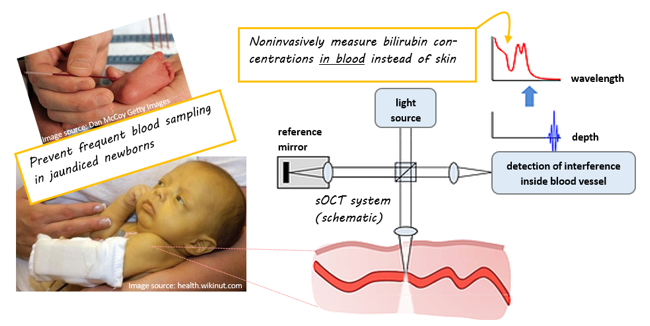
Premature and ill newborns are often subjected to invasive blood sampling. Invasive blood sampling is related to pain, stress, substantial blood loss and increased risk of infections – all factors that have been proven to impair the growth and development of this vulnerable patient group.
A possible alternative to invasive blood testing is optical spectroscopy: a fast, noninvasive technique, sensitive to the concentration dependent light absorption by two frequently – often exclusively – tested blood analytes in newborns (bilirubin and hemoglobin). Transcutaneous bilirubin meters (TcB meters) are devices based on optical spectroscopy, which are currently applied in the clinic to noninvasively measure bilirubin levels in the skin of newborns. This method is currently only used for screening purposes. TcB meters can prevent around 30% of all invasive blood samples for bilirubin determinations. For the other 70%, the technique is unfortunately not accurate enough to replace invasive blood sampling. The main reason is that the skin concentration of bilirubin is not directly related to the blood concentration (TSB), which is used for medical decision making.
At BMPI, we investigate how we can improve the clinical value of TcB meters - in close collaboration with the Isala Hospital. In parallel, we investigate how we can use another optical technique - optical coherence tomography - to potentially perform a more accurate noninvasive blood analysis.
Optical Coherence Tomography
Optical Coherence Tomography (OCT) is a high resolution, noninvasive 3D-imaging technique employing light. OCT is often referred to as the ‘optical analogue’ of ultrasound imaging. Similar to ultrasound imaging, OCT creates a cross sectional (or 3D) image of tissue from the depth resolved backscattered intensity. The major difference between OCT and ultrasound imaging, is that OCT uses light, instead of sound. This has the direct consequence that the resolution is many times better in OCT (in the order of one to several micrometers), but the imaging depth in tissue is lower (maximally 2 mm).

Image: OCT B-scans of muscular tissue (left) and fatty tissue (right).
OCT research at BMPI
At BMPI, we go beyond imaging with OCT. By investigating the spectroscopic content of the OCT signal at every depth inside the tissue of interest, we perform depth-resolved spectroscopy. Spectroscopy relates the wavelength dependent absorption and scattering of tissue to its chemical composition. As a result, we not only obtain structural information about the tissue of interest, but also chemical information.
This functional extension of OCT, called spectroscopic OCT (sOCT) has two major advantages compared to conventional optical spectroscopy: 1) it is localized, since we exactly know the depth from which our spectroscopic signal originates inside the tissue, and 2) it is quantitative, since we do not have make any assumptions on the optical path length that the light has traveled when calculating the optical absorption and/or scattering inside tissue.
We use visible sOCT to confine the spectroscopic measurement volume to blood only, for instance a single blood vessel. In that way, we exclude the background disturbance of extravasated bilirubin in the skin and measure the bilirubin concentration in blood directly. With that, we hope to achieve the highest possible measurement accuracy for noninvasive bilirubinometry, and reduce as many invasive blood samples as possible.
ALUMNI AND FORMER TEAM MEMBERS
PhD Students
- 2024 - Dr.ir. Alida Dam, PhD
- 2023 - Dr. Carlos Cuartas Vélez, PhD
- 2020 - Dr.ir. Colin Veenstra, PhD
Master Students
- 2023 - Rachel Foppen | Biomedical Engineering
- 2023 - Claudia Morsink | Biomedical Engineering
- 2021 - David Versteegen | Biomedical Engineering
- 2021 - Sander Logtenberg | Nano Technology
- 2019 - Saskia Kruitwagen | Biomedical Engineering
- 2018 - Marlijn van Erk | Biomedical Engineering
- 2016 - Ilse Runhart | Health Sciences
- 2016 - Lot Jeurink | Biomedical Engineering
Bachelor Students
- 2024 - Anna Karreman | Biomedical Engineering
- 2024 - Lotte Jonkman | Technical Medicine
- 2024 - Danee Rubingh | Technical Medicine
- 2024 - Verona Wigger | Technical Medicine
- 2024 - Jesse Schipper | Technical Medicine
- 2018 - Dafne Groener | Biomedical Engineering
- 2017 - Jeffrey Nagel | Applied Physics
- 2017 - David Versteegen | Biomedical Engineering
- 2017 - Saskia Yperlaan | Biomedical Engineering
- 2017 - Jelle Wouda | Mechanical Engineering - Saxion Hogeschool
- 2015 - Rianne van Dommelen | Biomedical Engineering
Interns
- 2025 - Karim Hassanu | Biomedical Engineering
- 2025 - Carmina Kaal | Biomedical Engineering
- 2023 - Isabella Gómez Gallego | Engineering Physics, Universidad EAFIT Colombia
- 2021 - Marleen Krommendijk | Biomedical Engineering
- 2019 - Elfi Hofmeijer | Biomedical Engineering
- 2019 - Nina Doorn | Technical Medicine
- 2019 - Stefan Lip | Technical Medicine
- 2019 - Nienke Timmermans | Technical Medicine
- 2019 - Leen Vanwinsen | Technical Medicine
- 2018 - Annelies Winkelhorst | Technical Medicine
SCIENTIFIC AWARDS
- Simon Stevin Gezel Award, Dutch Research Council (NWO)
- PhD thesis award, Dutch association for Biophysics and Biomedical Engineering (BIOPM)
- Tweelingprijs for best publication in the research field of Pediatrics, Maarten Kapelle foundation and the Dutch Association for Pediatrics (NVK)
- Best poster award, Photonics Cluster Netherlands at the Dutch Photonics Event
- Simon Stevin Student Award, Dutch Research Council (NWO)
Students who received awards on this project:
- Rosaline Mentink: Abbott Prize for best scientific internship in pediatrics, VU Medical Center
- Rosaline Mentink: Student Research Prize for best student research project, VU Medical Center
PUBLICATIONS - Transcutaneous bilirubin meters
- A.J. Dam-Vervloet, R. Foppen, C. Hulzebos, N. Bosschaart, Monte Carlo modeling of the influence of skin color on the delivered light dose in phototherapy for newborns with hyperbilirubinemia, Biophotonics Discovery 2(3), 032508 (2025), doi: https://doi.org/10.1117/1.BIOS.2.3.032508 (featured in SPIE News)
- A.J. Dam-Vervloet, C.F. Morskink, M.E. Krommendijk, I.M. Nijholt, H.L.M. van Straaten, L. Poot, N. Bosschaart, Skin color influences transcutaneous bilirubin measurements: a systematic in vitro evaluation, Pediatric Research (2024), doi: https://doi.org/10.1038/s41390-024-03081-y (selected for Science for Kids interview)
- A.J. Dam-Vervloet, F.A. de Boer, I.M. Nijholt, L. Poot, N. Bosschaart, H.L.M. van Straaten, Quantification of cephalocaudal progression of jaundice in preterm infants. Pediatric Research 94(1), 239-245 (2023), doi: https://doi.org/10.1038/s41390-022-02396-y
- C.F. Morsink, A.J. Dam-Vervloet, M.E. Krommendijk, M. Kaya, C. Cuartas-Vélez, T. Knop, K.J. Francis, N. Bosschaart, Design and characterization of color printed polyurethane films as biomedical phantom layers, Biomedical Optics Express 14(9), 4485-4506 (2023), doi: https://doi.org/10.1364/BOE.491695
- A.J. Dam-Vervloet, N. Bosschaart, H.L.M. van Straaten, L. Poot, C.V. Hulzebos, Irradiance footprint of phototherapy devices: a comparative study, Pediatric Research (2021), doi: https://doi.org/10.1038/s41390-021-01795-x, (Editor’s Focus)
- A.J. Dam-Vervloet, M.D van Erk, N. Doorn, S.G.J. Lip, N.A. Timmermans, L. Vanwinsen, F.A. de Boer, H.L.M. van Straaten, N. Bosschaart, Inter-device reproducibility of transcutaneous bilirubin meters, Pediatric Research 89, 770–775 (2021), doi: https://doi.org/10.1038/s41390-020-01118-6
- M.D. van Erk, A.J. Dam-Vervloet, F.A. de Boer, M.F. Boomsma, H. van Straaten, N. Bosschaart, How skin anatomy influences transcutaneous bilirubin determinations: an in vitro evaluation, Pediatric Research 86(4), 471-477 (2019), doi: https://doi.org/10.1038/s41390-019-0471-z, (Editor’s Focus)
- N. Bosschaart, J.H. Kok, A.M. Newsum, D.M. Ouweneel, R. Mentink, T.G. van Leeuwen, M.C.G. Aalders, Limitations and opportunities of transcutaneous bilirubin measurements, Pediatrics 129, 689-694 (2012), doi: 10.1542/peds.2011-2586
PUBLICATIONS - SPECTROSCOPIC OCT
- C. Cuartas-Vélez, I. Gómez Gallego, N. Bosschaart, In vivo quantification of total hemoglobin concentrations in healthy human subjects with visible-light spectroscopic optical coherence tomography, Biomedical Optics Express 16(4), 1569-1581 (2025), doi: https://doi.org/10.1364/BOE.549671
- A.S. Purandare, C. Cuartas-Vélez, N. Smeman, M. Schremb, N. Bosschaart, S. Vanapalli, Experimental and theoretical investigation of the Leidenfrost dynamics of solid carbon dioxide discs sublimating on a solid substrate, International Journal of Heat and Mass Transfer 224, 125300 (2024), doi: https://doi.org/10.1016/j.ijheatmasstransfer.2024.125300
- C. Cuartas-Vélez, H.H.T. Middelkamp, A.D. van der Meer, A. van den Berg, N. Bosschaart, Tracking the dynamics of thrombus formation in a blood vessel-on-chip with visible-light optical coherence tomography, Biomedical Optics Express 14(11), 5642-5655 (2023), doi: https://doi.org/10.1364/BOE.500434
- C. Cuartas-Vélez, C. Veenstra, S. Kruitwagen, W. Petersen, N. Bosschaart, Optical density based quantification of total haemoglobin concentrations with spectroscopic optical coherence tomography, Scientific Reports 11(1), 1-10 (2021), doi: https://doi.org/10.1038/s41598-021-88063-4
- Y.B. Arik, W. Buijsman, J. Loessberg-Zahl, C. Cuartas-Vélez, C. Veenstra, S. Logtenberg, A.M. Grobbink, P. Bergveld, G. Gagliardi, A.I. den Hollander, N. Bosschaart, A. van den Berg, R. Passier, A.D. van der Meer, Microfluidic organ-on-a-chip model of the outer blood-retinal barrier with clinically relevant read-outs for tissue permeability and vascular structure, Lab on a Chip 21(2), 272-283 (2020), doi: https://doi.org/10.1039/D0LC00639D
- C. Veenstra, D. Every, W. Petersen, J.B. van Goudoever, W. Steenbergen, N. Bosschaart, Dependency of the optical scattering properties of human milk on casein content and common sample preparation methods, Journal of Biomedical Optics 25(4), 045001 (2020), doi: https://doi.org/10.1117/1.JBO.25.4.045001
- C. Veenstra, S. Kruitwagen, D. Groener, W. Petersen, W. Steenbergen, N. Bosschaart, Quantification of total haemoglobin concentrations in human whole blood by spectroscopic visible-light optical coherence tomography, Scientific Reports 9(1), 1-8 (2019), doi: https://doi.org/10.1038/s41598-019-51721-9
- C. Veenstra, A. Lenferink, W. Petersen, W. Steenbergen, N. Bosschaart, Optical properties of human milk, Biomedical Optics Express 10(8), 4059-4074 (2019), doi: https://doi.org/10.1364/BOE.10.004059
- C. Veenstra, W. Petersen, I.M. Vellekoop, W. Steenbergen, N. Bosschaart, Spatially confined quantification of bilirubin concentrations by spectroscopic visible-light optical coherence tomography, Biomedical Optics Express 9(8), 3581-3589 (2019), doi: https://doi.org/10.1364/BOE.9.003581
- N. Bosschaart, T.G. van Leeuwen, M.C.G. Aalders, D.J. Faber, Quantitative comparison of analysis methods for spectroscopic optical coherence tomography, Biomedical Optics Express 4, 2570-2584 (2013), doi: https://doi.org/10.1364/BOE.4.002570
- N. Bosschaart, M.C.G. Aalders, T.G. van Leeuwen, D.J. Faber, Spectral domain detection in low-coherence spectroscopy, Biomedical Optics Express 3, 2263-2272 (2012), doi: https://doi.org/10.1364/BOE.3.002263
- N. Bosschaart, D.J. Faber, T.G. van Leeuwen, M.C.G. Aalders, In vivo low-coherence spectroscopic measurements of local hemoglobin absorption spectra in human skin, Journal of Biomedical Optics 16, 100504 (2011), doi: https://doi.org/10.1117/1.3644497
- N. Bosschaart, D.J. Faber, T.G. van Leeuwen, M.C.G. Aalders, Measurements of wavelength dependent scattering and backscattering coefficients by low-coherence spectroscopy, Journal of Biomedical Optics 16, 030503 (2011), https://doi.org/10.1117/1.3553005
- N. Bosschaart, M.C.G. Aalders, D.J. Faber, J.J.A. Weda, M.J.C. van Gemert, T.G. van Leeuwen, Quantitative measurements of absorption spectra in scattering media by low-coherence spectroscopy, Optics Letters 34, 3746-3748 (2009), https://doi.org/10.1364/OL.34.003746
