
Research Technician
E-mail: A.Mentink@utwente.nl
Telephone: +31 53 489 3355
Fax: +31 53 489 3511
Address: Faculty of Science and Technology
Medical Cell BioPhysics (MCBP)
Building: Carré
Room: CR4538
Drienerlolaan 5
P.O. Box 217
7500 AE Enschede
The Netherlands
Project goals
The CANCER-ID project aims to validate technologies for Circulating Tumor Cells (CTCs), circulating free DNA (cfDNA) and circulating cell free microRNAs (cfmiRs) as blood-based biomarkers to determine the absence/presence of drug targets and assess response to treatment. The two cancer types that will be investigated are Non-Small Cell Lung Cancer and Her2-therapy resistant metastatic breast cancer.
Project background
Circulating Tumor Cells (CTCs) are cells that break free from the tumor and circulate in the blood of cancer patients, forming metastases. Furthermore, blood plasma may contain circulating tumor DNA. The current standard for CTC detection is the FDA-cleared CellSearch (CS) and this will be used as the “golden standard” in CANCER-ID. Not all CTCs are detected by CS and CTC detection will be improved by broadening the reagents for detection and exploring the presence of CTCs in the blood discarded by the CS with new approaches.
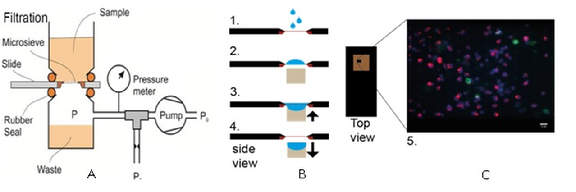
Figure 1. A) Blood discarded by the CS system will be filtered through a 5 µM microsieve. B) Cells will be captured in the pores and a staining can be performed. C) Commonly a nuclear stain (blue) is used in combination with cytokeratin markers (green) and CD45 (red) to distinguish tumor cells from white blood cells.
The increasing number of treatment options for patients with metastatic cancer has created an accompanying need for biomarkers to determine if the tumor will be responsive to the intended therapy, to monitor drug efficacy and to anticipate emergent drug resistance. Ideally, these biomarkers would be obtained by minimally invasive means to allow serial sampling enabling quantitative real-time molecular analyses of tumor heterogeneity, evolution and drug responsiveness. When implemented in clinical practice it will result in personalized medicine by adapting therapies to the individual risk of the cancer patient.
CD45 depletion
Commonly, blood volumes of 1–10 mL are analyzed and in some cases these volumes are too low to detect rare CTCs. Diagnostic LeukApheresis (DLA) is a standard clinical method that is frequently used to isolate mononuclear cells (MNCs) from blood for various applications. DLA is an extracorporeal therapy in which the blood of a donor or patient is passed through an apparatus that continuously performs density-based cell separation of MNCs from several liters of total processed blood volume and returns the remainder to the circulation. The clinical value of DLA, which is more invasive than a simple venipuncture, is clearly related to the further molecular and functional characterization of CTCs in the context of personalized molecular therapies. To isolate CTCs from a leukapheresis product the white blood cells need to be depleted. For this purpose we will test multiple antibody-based CD45 depletion assays and compare performance.
